Antibodies Can Selectively Shut Down Harmful T Cells Without Weakening the Entire Immune System
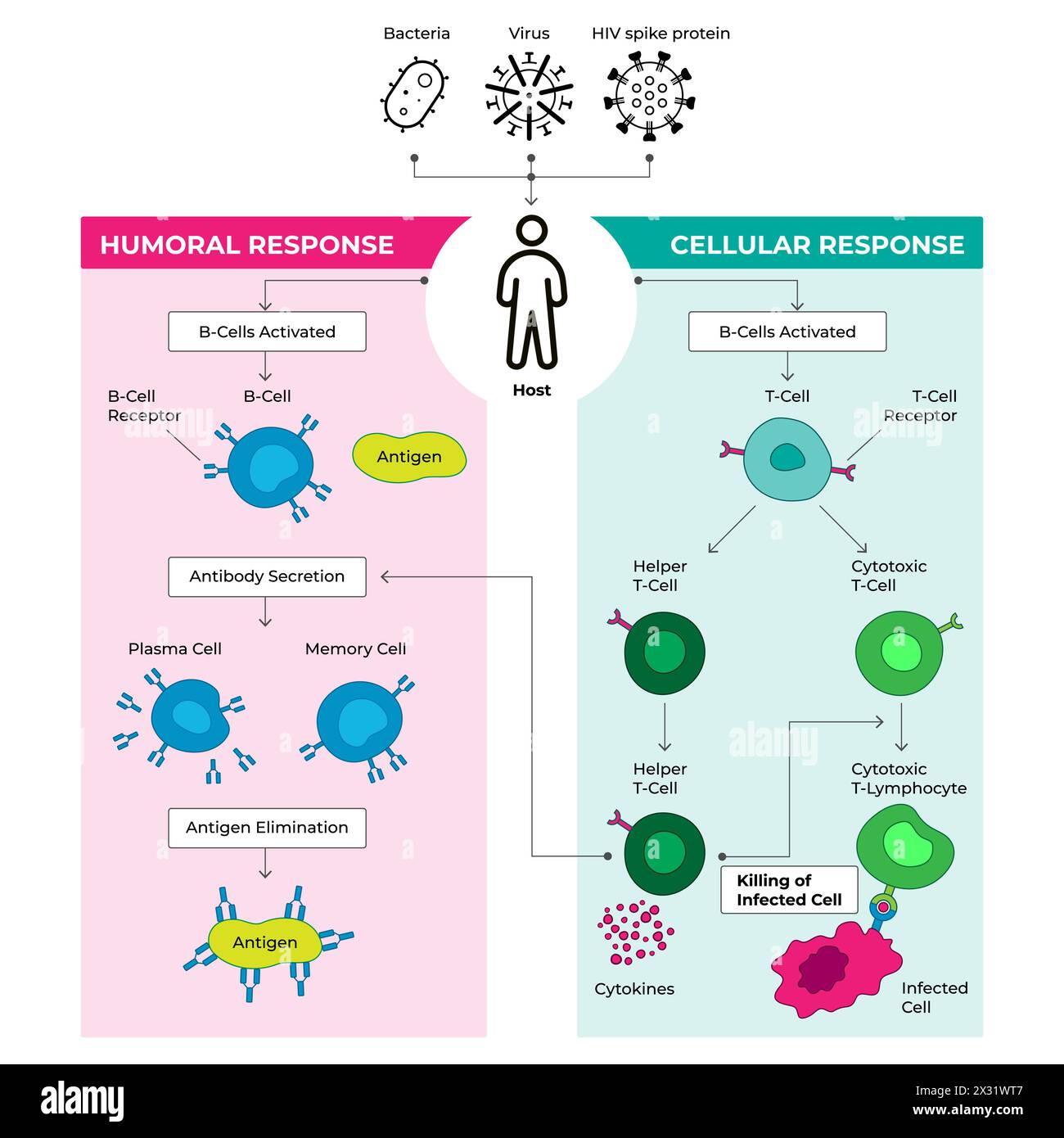
- Researchers have developed a novel antibody that can selectively disable harmful T cells while leaving the rest of the immune system intact, according to a report published by...
- The antibody works by targeting specific overactive T cells that contribute to autoimmune responses, effectively shutting them down at their source.
- This development builds on ongoing efforts to create more precise immunotherapies that avoid the drawbacks of current treatments, which often weaken the entire immune system and increase susceptibility...
Researchers have developed a novel antibody that can selectively disable harmful T cells while leaving the rest of the immune system intact, according to a report published by Medical Xpress. The breakthrough offers a potential new approach for treating autoimmune diseases without compromising the body’s ability to fight infections.
The antibody works by targeting specific overactive T cells that contribute to autoimmune responses, effectively shutting them down at their source. Unlike broader immunosuppressive therapies, this method aims to preserve overall immune function while addressing the root cause of harmful inflammation.
This development builds on ongoing efforts to create more precise immunotherapies that avoid the drawbacks of current treatments, which often weaken the entire immune system and increase susceptibility to infections. By focusing exclusively on pathogenic T cells, the antibody could provide a safer alternative for conditions such as rheumatoid arthritis, lupus, and multiple sclerosis.
According to the research, the antibody demonstrates selectivity in preclinical models, distinguishing between harmful and beneficial T cells based on their activation state and antigen specificity. This precision allows it to modulate immune activity without triggering widespread immunosuppression.
Experts note that while the findings are promising, the technology remains in early stages of development. Further studies will be needed to confirm its safety and efficacy in humans, including clinical trials to evaluate long-term effects and potential off-target impacts.
The approach represents a shift toward more targeted immunomodulation, aligning with broader trends in medical research to treat complex immune disorders with greater specificity. If successful in human applications, such therapies could transform how autoimmune diseases are managed, reducing reliance on broad-spectrum drugs that carry significant side effects.
As research progresses, scientists will continue to investigate the mechanisms underlying T cell regulation and explore ways to refine antibody-based interventions for clinical use. The goal remains to achieve durable remission in autoimmune conditions while maintaining protective immunity against pathogens.
