AcTau174: A Novel CSF Biomarker for FTLD-TDP Diagnosis and Progression
- Researchers have developed a new ultrasensitive immunoassay to quantify acetylated tau at lysine 174 (AcTau174) in cerebrospinal fluid (CSF), providing a method to distinguish between different underlying pathologies...
- Frontotemporal dementia (FTD) comprises a group of clinically heterogeneous syndromes characterized by impairments in language, executive function, and behavior.
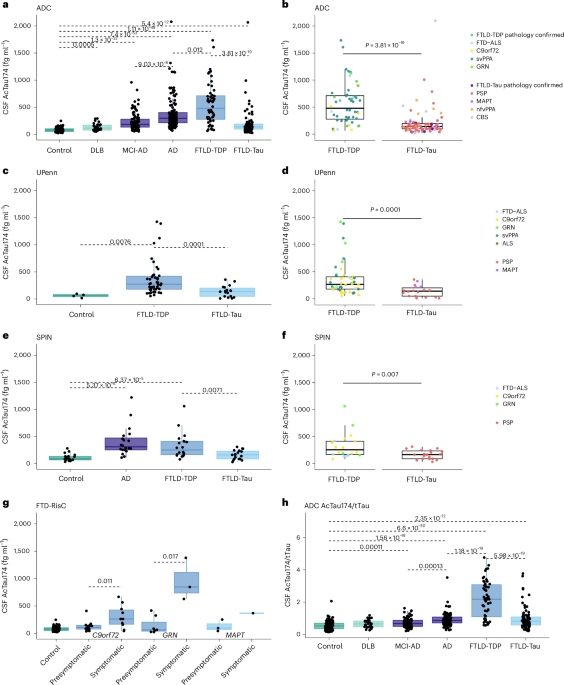
- In a sporadic cohort of 513 participants, AcTau174 concentrations were found to be higher in all dementia groups compared to controls, including those with FTLD-Tau, Alzheimer’s disease (AD),...
Researchers have developed a new ultrasensitive immunoassay to quantify acetylated tau at lysine 174 (AcTau174) in cerebrospinal fluid (CSF), providing a method to distinguish between different underlying pathologies in patients with frontotemporal lobar degeneration (FTLD). According to a study published in Nature Medicine on April 15, 2026, this biomarker is elevated in FTLD associated with TAR DNA-binding protein (FTLD-TDP) and can effectively differentiate it from FTLD associated with tau pathology (FTLD-Tau).
Frontotemporal dementia (FTD) comprises a group of clinically heterogeneous syndromes characterized by impairments in language, executive function, and behavior. Identifying whether the underlying pathology is driven by tau or TDP-43 is critical for advancing clinical trials that target these specific pathologies.
Diagnostic Accuracy of AcTau174
In a sporadic cohort of 513 participants, AcTau174 concentrations were found to be higher in all dementia groups compared to controls, including those with FTLD-Tau, Alzheimer’s disease (AD), mild cognitive impairment (MCI)-AD, and dementia with Lewy bodies (DLB). However, the most significant increase was observed in the FTLD-TDP group.
The biomarker demonstrated high accuracy in discriminating FTLD-TDP from other groups. In the discovery cohort, the area under the curve (AUC) for discriminating FTLD-TDP from FTLD-Tau was 0.83, with a 95% confidence interval of 0.75–0.91. When contrasting FTLD-TDP against controls, the AUC was 0.95, with a 95% confidence interval of 0.92–0.99.
These results were replicated in independent validation cohorts consisting of 164 patients and 24 controls. While the accuracy in distinguishing FTLD-TDP from FTLD-Tau was somewhat lower in these validation cohorts, with an AUC range of 0.75–0.79, the findings remained consistent.
The study noted that the largest increases in AcTau174 were particularly evident in patients with semantic variant primary progressive aphasia (svPPA) and those who are carriers of the GRN mutation.
Prognostic Value and Disease Progression
Beyond its diagnostic utility, AcTau174 appears to serve as a prognostic marker. The research indicates that higher concentrations of the biomarker are associated with a faster rate of cognitive decline over time.
This association with disease progression was observed across multiple groups, including those with FTLD-TDP, AD, and MCI-AD. In patients with FTLD-TDP specifically, the signal in the spinal fluid aligned with the severity of the disease, with higher levels correlating to more rapid declines in thinking and language abilities.
Impact on Clinical Trials and Care
The lack of fluid biomarkers and the inherent clinical heterogeneity of FTD have historically limited the effectiveness of clinical trials. The introduction of AcTau174 may address these challenges by aiding in the identification of patients at risk of rapid decline.
The ability to accurately separate TDP-43 driven disease from tau-driven disease in living patients could support trial enrichment, allowing researchers to select the most appropriate patients for targeted interventions. This reduction in diagnostic guesswork may lead to faster and more confident diagnoses for families.
CSF AcTau174 has great potential to discriminate FTLD-TDP from FTLD-Tau as a biomarker reflecting FTLD-TDP disease severity and progression.
Nature Medicine
While the results are described as promising, the researchers indicated that further checks are still required to fully establish the utility of the biomarker in clinical practice.
