Antibody-Based Apheresis Shows Promise in Treating Very Preterm Preeclampsia
- A first-in-human pilot trial has demonstrated that selectively removing a key placental protein from the blood of women with very preterm preeclampsia is safe and may modestly lower...
- The open-label, single-arm trial enrolled 16 women diagnosed with very preterm preeclampsia, defined as occurring before 32 weeks of gestation.
- In the first phase of the trial (Phase A), seven women received single ascending doses of the treatment.
A first-in-human pilot trial has demonstrated that selectively removing a key placental protein from the blood of women with very preterm preeclampsia is safe and may modestly lower blood pressure while extending pregnancy, according to findings published on April 27, 2026, in Nature Medicine. The study, which tested an antibody-based apheresis technique, provides early evidence that targeting soluble Fms-like tyrosine kinase 1 (sFlt-1) could offer a disease-specific treatment for preeclampsia, a life-threatening pregnancy complication for which no targeted therapies currently exist.
The Trial’s Design and Key Findings
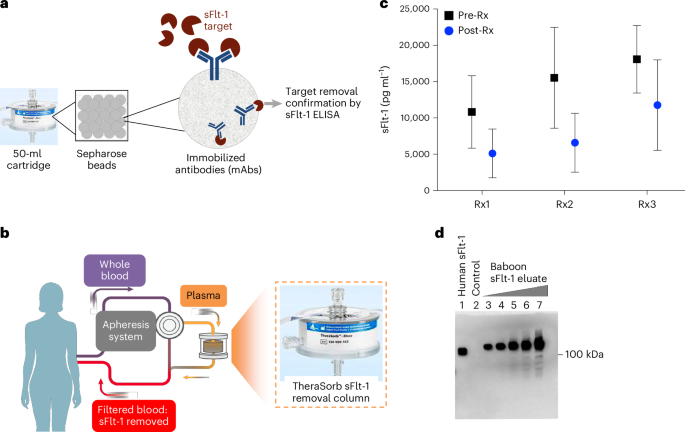
The open-label, single-arm trial enrolled 16 women diagnosed with very preterm preeclampsia, defined as occurring before 32 weeks of gestation. Participants underwent extracorporeal apheresis using an adsorber containing high-affinity IgG1 antibodies designed to bind and remove circulating sFlt-1, a protein secreted by the placenta that is strongly implicated in the development of preeclampsia. The primary endpoints of the study were safety and tolerability, with secondary assessments of blood pressure changes, pregnancy prolongation, and fetal outcomes.

In the first phase of the trial (Phase A), seven women received single ascending doses of the treatment. Pre-apheresis sFlt-1 levels averaged 15,120 ± 4,484 pg/ml. During and after the procedure, maternal and fetal vital signs remained stable, and umbilical artery pulsatility indices—an indicator of placental blood flow—showed no adverse changes. In the second phase (Phase B), nine women with a median gestational age of 30.3 weeks (interquartile range: 29.3–30.9 weeks) received multiple apheresis sessions. These participants had elevated blood pressure at baseline, with systolic and diastolic pressures averaging 146 ± 10 mmHg and 92 ± 5 mmHg, respectively, and pre-apheresis sFlt-1 levels of 11,960 ± 3,056 pg/ml.
Each apheresis session reduced circulating sFlt-1 levels by an average of 16.7 ± 7.6% and lowered mean arterial pressure by 4.1 ± 7.8 mmHg. The reductions in blood pressure strongly correlated with decreases in sFlt-1 levels (Spearman’s correlation coefficient, R = 0.63), suggesting a direct link between the protein’s removal and clinical improvement. Pregnancy was prolonged by a median of 10 days (range: 3–19 days) from the time of admission, and neonatal birth weights either remained stable or increased among those with the longest extensions of pregnancy.
Why sFlt-1 Matters in Preeclampsia
Preeclampsia affects approximately 5–8% of pregnancies worldwide and is a leading cause of maternal and fetal morbidity and mortality. The condition is characterized by new-onset hypertension and organ dysfunction, often involving the kidneys, liver, or brain. While the exact mechanisms remain incompletely understood, sFlt-1 is widely recognized as a central mediator. The protein acts as an anti-angiogenic factor, binding to and neutralizing vascular endothelial growth factor (VEGF) and placental growth factor (PlGF), which are essential for maintaining healthy blood vessel function in the placenta and maternal endothelium.
Elevated levels of sFlt-1 are associated with endothelial dysfunction, vasoconstriction, and the clinical symptoms of preeclampsia, including hypertension and proteinuria. Current management strategies focus on symptom control—such as antihypertensive medications and magnesium sulfate to prevent seizures—and delivery of the baby, which remains the only definitive cure. However, early delivery, particularly before 34 weeks of gestation, carries significant risks for the newborn, including respiratory distress syndrome, intraventricular hemorrhage, and long-term neurodevelopmental challenges.
The trial’s findings build on earlier research exploring sFlt-1 as a therapeutic target. A 2015 study published in the Journal of the American Society of Nephrology demonstrated that dextran sulfate apheresis could reduce sFlt-1 levels and proteinuria in women with very preterm preeclampsia, with pregnancy prolongation observed in some cases. However, that approach was less selective, also removing lipids and other molecules from the blood. The current trial’s use of antibody-based apheresis offers a more targeted mechanism, potentially minimizing off-target effects while directly addressing the underlying pathophysiology of preeclampsia.
Safety and Limitations
The trial reported no major adverse maternal or fetal events associated with the apheresis procedure. Maternal vital signs and fetal monitoring parameters remained stable throughout the treatment, and no participants experienced complications such as placental abruption, pulmonary edema, or severe allergic reactions. However, the authors emphasized that the study was not designed to assess efficacy definitively. As a single-arm trial without a placebo or control group, the observed benefits—such as blood pressure reductions and pregnancy prolongation—cannot be attributed solely to the intervention without further investigation.
the trial’s small sample size (16 participants) limits the generalizability of the findings. The authors noted that larger, randomized controlled trials are necessary to confirm the safety and efficacy of the approach, as well as to determine optimal dosing, treatment frequency, and long-term outcomes for both mothers and infants. The study also did not address whether the intervention could be effective in less severe cases of preeclampsia or in women at earlier stages of the disease.
Broader Implications and Next Steps
The trial’s results represent a step toward a disease-modifying treatment for preeclampsia, a condition that has long lacked targeted therapeutic options. If validated in larger studies, antibody-based apheresis could provide a bridge to safer delivery for women with very preterm preeclampsia, potentially reducing the need for early delivery and improving neonatal outcomes. The approach may also offer insights into the broader role of sFlt-1 in pregnancy-related disorders and other conditions characterized by endothelial dysfunction.

Researchers are already planning follow-up studies to address the current trial’s limitations. A proof-of-concept trial (NCT02923206) evaluating the TheraSorb sFlt-1 adsorber, a similar device, is underway, with results expected to provide additional data on the intervention’s safety and potential benefits. Future research may also explore whether combining apheresis with other therapies, such as antihypertensive medications or anti-inflammatory agents, could enhance outcomes.
For now, the findings underscore the need for continued investment in preeclampsia research. As the authors of the Nature Medicine study concluded, “The treatment supports conducting controlled trials to evaluate its safety and efficacy at a larger scale.” Until such trials are completed, the standard of care for preeclampsia remains delivery of the baby, with all the attendant risks of prematurity.
What In other words for Patients and Clinicians
For women diagnosed with very preterm preeclampsia and their healthcare providers, the trial offers a glimmer of hope but also a reminder of the complexity of the condition. While the results are promising, the intervention is not yet available outside of clinical trials, and its long-term safety and efficacy remain unproven. Patients and clinicians should continue to rely on established management protocols, including close monitoring, blood pressure control, and timely delivery when indicated.
Clinicians may view the trial as a validation of the growing body of evidence supporting sFlt-1 as a therapeutic target in preeclampsia. However, they should also counsel patients that the intervention is experimental and that participation in clinical trials remains the only way to access such treatments at this stage. For researchers, the study highlights the potential of precision medicine approaches in obstetrics, where targeted therapies have historically been limited.
As the field advances, the integration of biomarkers like sFlt-1 and PlGF into clinical practice may improve risk stratification and guide treatment decisions. For example, women with particularly high sFlt-1 levels might be identified as candidates for future interventions, while those with lower levels could be managed with standard care. Such personalized approaches could help balance the risks of prematurity with the dangers of prolonging a high-risk pregnancy.
the Nature Medicine trial represents a critical step in the journey toward a targeted therapy for preeclampsia. While the road ahead is long, the study’s findings provide a foundation for future research and a potential path to transforming the management of one of pregnancy’s most dangerous complications.
