Cardiotropic AAV Gene Therapy for Heart Failure: Phase 1 Trial
- For individuals battling the debilitating effects of NYHA class III systolic heart failure, a new study offers a glimmer of hope.
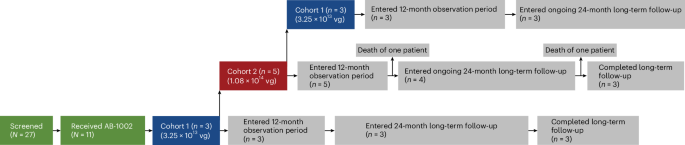
- The study, registered under ClinicalTrials.gov as NCT04179643, followed a dose-escalation approach.
- The first group of patients received a dose of 3.25 × 10^13 vg (viral genomes) of AB-1002. Subsequent groups were slated to receive higher doses.
Hope on the Horizon: Gene Therapy Shows Promise for Heart Failure Patients
For individuals battling the debilitating effects of NYHA class III systolic heart failure, a new study offers a glimmer of hope. Researchers have been exploring the safety and potential benefits of AB-1002, a novel gene therapy, delivered directly into the heart via an intracoronary infusion. This phase 1, open-label study, conducted across four sites in the USA (Minnesota, Ohio, and Wisconsin), focused on patients with nonischemic cardiomyopathy and a specific range of heart function (LVEF of 15-35%).
The Study Design:
The study, registered under ClinicalTrials.gov as NCT04179643, followed a dose-escalation approach. This means that researchers started with a low dose of AB-1002 and gradually increased it in subsequent groups of patients,carefully monitoring for any adverse effects. The initial plan involved three dose levels, with safety being the paramount concern. A clinical safety committee, comprised of the study’s principal investigators and a cardiologist, meticulously reviewed the data to ensure patient well-being.
The first group of patients received a dose of 3.25 × 10^13 vg (viral genomes) of AB-1002. Subsequent groups were slated to receive higher doses. Though, based on early efficacy signals in the first group, coupled with some observed elevations in liver enzymes and the progress of antibodies against the viral vector, the study protocol was adjusted. Instead of escalating to higher doses, the researchers decided to enroll three additional patients in the initial dose group (3.25 × 10^13 vg) to gather more data at that level.
Who Participated?
The study enrolled men and women over 18 years of age who met specific criteria. these included:
* Diagnosis of chronic nonischemic cardiomyopathy.
