Chirality Shock: Mirror-Proof Drugs Created by Geneva Chemists
The Double-Edged Sword of Chirality: A Breakthrough in Stable Drug Design
Imagine a molecule that could save a life,yet its identical twin is a deadly poison. This seemingly paradoxical reality is the essence of “chirality,” a basic concept in chemistry that dictates how molecules interact with our bodies. Much like our left and right hands, chiral molecules share the same atomic composition but differ in their three-dimensional arrangement, a subtle distinction that can have profound biological consequences. For decades,controlling this molecular asymmetry has been a cornerstone of effective drug design. Now, a groundbreaking study from the University of Geneva (UNIGE), in collaboration with the University of Pisa, introduces a new family of remarkably stable chiral molecules, heralding a new era for geometry-controlled pharmaceuticals.
Chirality: The Molecular Handshake
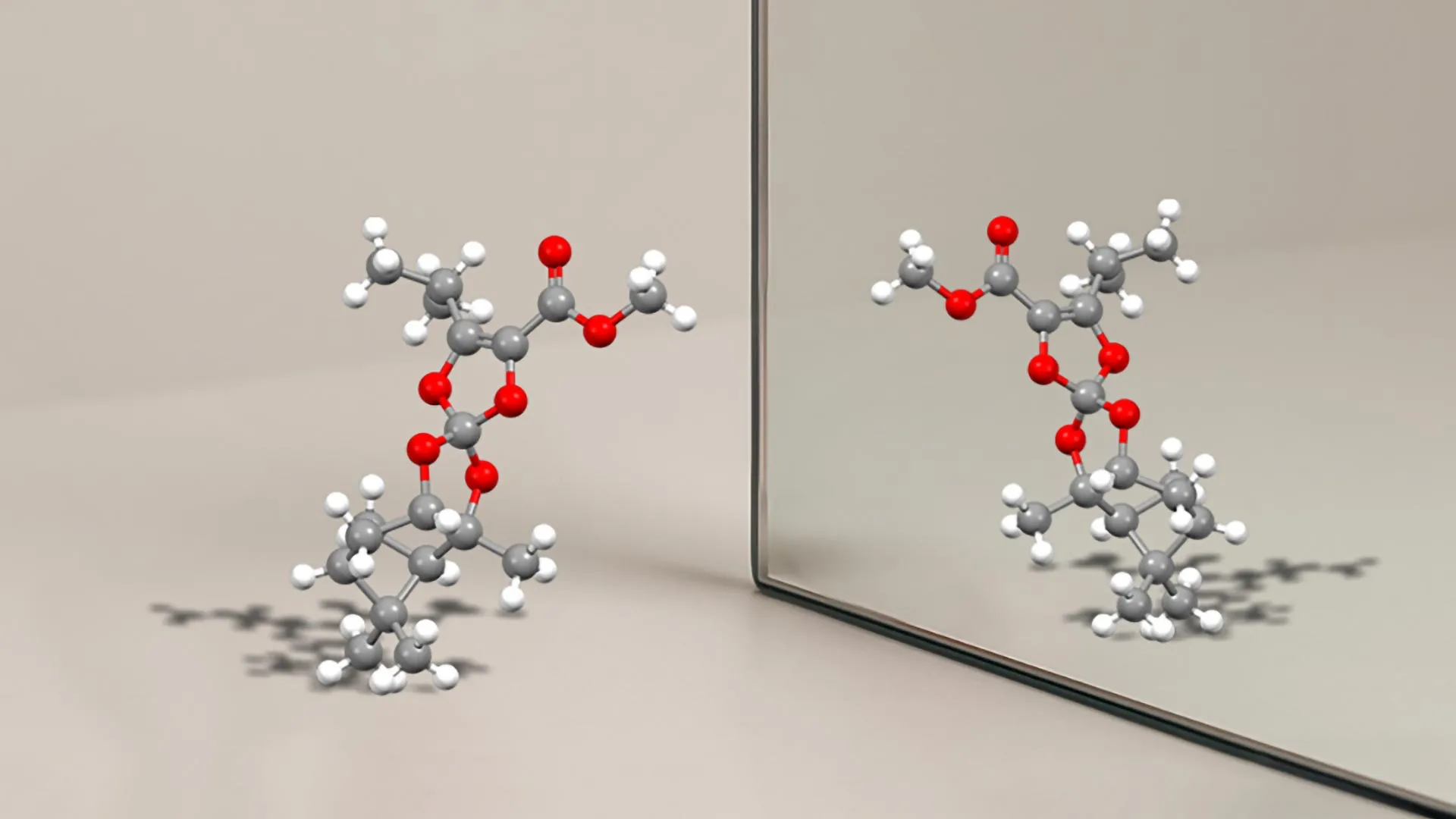
At its core, chirality describes objects that are non-superimposable on their mirror images. Think of your hands: they are mirror images, but no amount of twisting or turning will make your left hand perfectly align with your right. In the molecular world, this asymmetry often arises from a “stereogenic center,” typically a carbon atom bonded to four different groups. This precise spatial arrangement is critical for how molecules interact with biological targets, such as proteins and enzymes, which are themselves chiral. A drug molecule needs to “fit” its target like a key in a lock, and only the correct chiral form will achieve this precise interaction.
A Novel Stereogenic Center: A Chemical First
The UNIGE team, led by professor Jérôme lacour, has achieved a significant feat by creating a new type of stereogenic center. Unlike conventional chiral centers that rely on carbon atoms bonded to carbon chains, this novel center features a central carbon atom surrounded by oxygen and nitrogen atoms. This represents a pioneering achievement in chemistry, as molecules incorporating this specific arrangement had never before been isolated in a stable form.
“molecules with this new type of stereogenic center had never before been isolated in a stable form,” explains Professor Lacour. ”Their synthesis and characterization mark a major conceptual and experimental breakthrough.”
Unprecedented Stability: The key to Safety
The stability of chiral molecules is paramount in pharmaceutical applications. The inherent similarity between mirror-image molecules means they can, under certain conditions like temperature fluctuations, spontaneously interconvert. This “switching” could transform a life-saving drug into an inactive or even toxic substance.The newly developed chiral molecules from UNIGE exhibit exceptional stability, making such interconversion highly improbable.Olivier Viudes, a PhD student and the study’s first author, elaborates on this remarkable property: “Using dynamic chromatography techniques and quantum chemistry calculations, we have shown that, for the first molecule developed, it would take 84,000 years at room temperature for half a sample to transform into its mirror molecule.” For a second molecule, this conversion time was estimated at 227 days at 25°C. This extraordinary stability ensures the integrity of the drug during storage and use, eliminating the need for specialized handling conditions.
New Frontiers in Drug Design and Materials Science
The implications of these stable, geometry-controlled chiral molecules are far-reaching. They provide chemists with a novel toolkit for precisely organizing molecular space, opening up new avenues for drug discovery and the development of advanced materials.
“These novel stereogenic centers offer a new way of organizing molecular space,” concludes Gennaro Pescitelli, professor at the University of Pisa and co-principal investigator of the article.”They open up a whole new degree of freedom and imagination in chemical synthesis.”
This breakthrough promises to enhance the safety and efficacy of future pharmaceuticals and pave the way for innovative materials with tailored properties, all thanks to a deeper understanding and control of molecular handedness.
