Gene-Edited Pig Liver Sustains Human for 171 Days – Xenotransplantation Breakthrough
- In a landmark achievement for the field of xenotransplantation, a patient in China survived for 171 days after receiving a partially functioning pig liver, marking the longest survival...
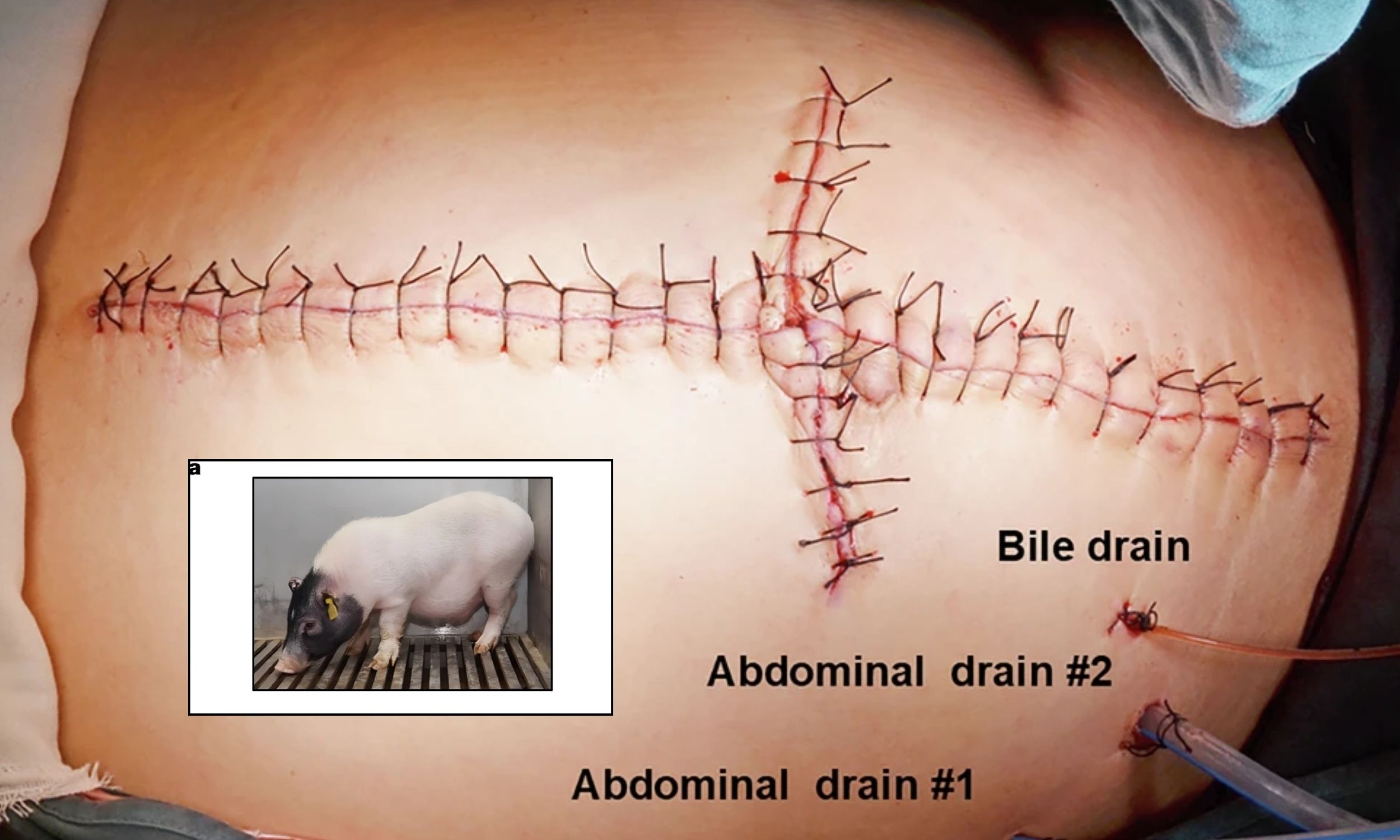
- The experimental procedure involved placing the gene-edited pig liver alongside the patient’s own failing liver, allowing both organs to function in parallel.
- The recipient, suffering from advanced liver cancer, faced a grim prognosis with limited options for standard treatments.
In a landmark achievement for the field of xenotransplantation, a patient in China survived for after receiving a partially functioning pig liver, marking the longest survival to date for a recipient supported by a pig organ. The breakthrough, documented by clinicians at the First Affiliated Hospital of Anhui Medical University (AHMU), offers a potential bridge for individuals awaiting human liver transplants, though significant hurdles remain.
A Temporary Support System
The experimental procedure involved placing the gene-edited pig liver alongside the patient’s own failing liver, allowing both organs to function in parallel. This approach was deliberately designed as a reversible support system, rather than a permanent replacement, limiting the extent of the experiment while providing crucial time for the patient’s native liver to potentially recover. Researchers closely monitored the metabolic functions of both organs, noting stable liver and kidney markers during the initial post-surgery.
A Patient in Need
The recipient, suffering from advanced liver cancer, faced a grim prognosis with limited options for standard treatments. Surgeons hoped the additional liver tissue would provide essential metabolic support, allowing the patient’s remaining liver to regain strength. The procedure was undertaken with informed consent from both the patient and his daughter, recognizing the compassionate use nature of the treatment rather than routine clinical care. It’s important to note that the success in this single case does not guarantee similar outcomes for all transplant candidates.
The Role of Gene Editing
A major obstacle to xenotransplantation is the human body’s natural rejection of foreign tissue. To overcome this, the donor pig underwent extensive gene editing. Ten targeted changes were made to remove pig genes that trigger human antibody responses and to add human versions of proteins that regulate inflammation, and clotting. Even with these modifications, the risk of rejection and other immune-related complications remains a significant concern, requiring careful monitoring.
Early Signs of Functionality
The transplanted pig liver demonstrated early signs of functionality, a critical indicator of success. Within two hours of implantation in a separate study involving a brain-dead recipient, the six-gene-edited pig liver began producing bile, a crucial digestive fluid. Before removal, the liver had produced of bile and showed an increase in albumin levels, a protein essential for maintaining fluid balance. This demonstrated the liver’s capacity to perform essential metabolic functions.
Addressing Thrombotic Microangiopathy
The trial encountered its most significant challenge when small blood vessels within the pig liver began to clog, a condition known as thrombotic microangiopathy. This complication, linked to immune-driven inflammation, led to the removal of the pig liver on . Following removal, doctors employed antibody-blocking therapy and blood-filtering treatments to mitigate the inflammatory response. This experience highlighted the importance of addressing vessel health as a critical factor for future success.
Ensuring Donor Pig Safety
Beyond the risk of rejection, concerns exist regarding the potential transmission of porcine viruses to humans. To mitigate this risk, donor pigs are raised in biosecure facilities and rigorously screened for viruses that could pose a threat to human health. In the case of the brain-dead recipient, researchers detected no evidence of pig viruses in blood or liver samples. However, federal guidance emphasizes the need for lifelong monitoring, as some infections may not manifest for months or years.
Managing Immune Suppression
Suppressing the immune system is crucial to prevent rejection of the transplanted organ. However, immunosuppressant drugs also increase the risk of infection. Researchers carefully monitored immune cell counts and adjusted treatment regimens to strike a balance between preventing rejection and maintaining sufficient immunity. Future trials will need to refine this balance to optimize patient safety.
A Bridge to Transplant
Currently, researchers view pig livers as a temporary bridge to keep patients stable until a human organ becomes available. With over people on the United States transplant waitlist, even a few extra months of support could be life-saving. This study represents the first clear demonstration that a pig-derived liver can function within a human body and potentially substitute for a failing human liver.
Future Steps and Considerations
Before pig liver transplants become widely available, further research is needed to replicate these results and manage predictable risks. Future studies will require clear protocols for graft removal and contingency plans for securing human organs. Researchers are initially focusing on patients with unresectable liver cancer and acute liver failure, where treatment options are limited. If these trials prove successful, larger-scale programs may be considered, but stringent safeguards regarding clotting and infection will remain paramount.
This single patient case demonstrates the potential of pig livers to provide meaningful support in humans, even if long-term survival remains a challenge. Prolonged survival will depend on overcoming clotting problems, carefully selecting appropriate patients, and implementing long-term follow-up monitoring. The findings of this study have been published in Nature.
