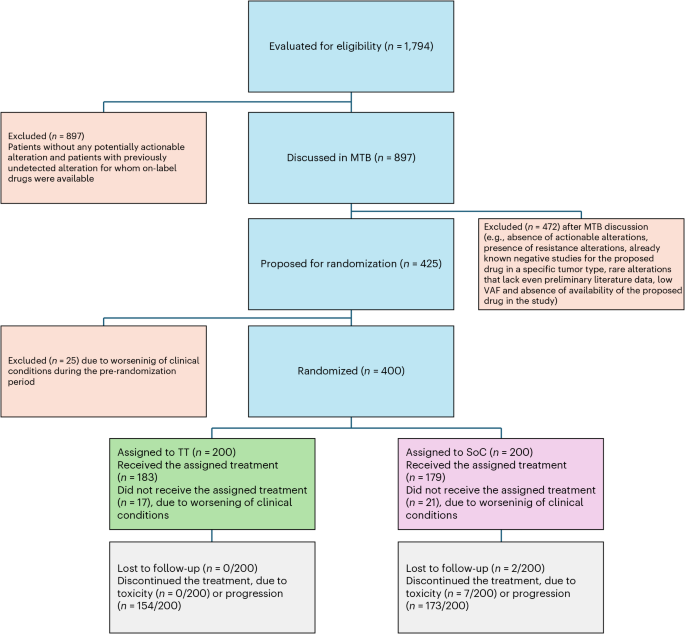
Genomic-Matched Therapy in Advanced Solid Tumors: ROME Trial Results
- Okay, here's a breakdown of the key information from the provided text, organized for clarity.This summarizes the study's methods:
- * Targeted Therapy (TT): Treatment was selected based on specific pathway, signature, or gene alteration identified in the patient's tumor (details in Extended Data Table 4).
- * Primary Endpoint: Overall Response Rate (ORR) - the percentage of patients who had a complete or partial response to treatment.
Okay, here’s a breakdown of the key information from the provided text, organized for clarity.This summarizes the study’s methods:
1. Treatments:
* Targeted Therapy (TT): Treatment was selected based on specific pathway, signature, or gene alteration identified in the patient’s tumor (details in Extended Data Table 4). Generally preferred if safety data was available.
* Standard of Care (SoC): The standard treatment for the patient’s cancer type.
2. Study Endpoints (What they where measuring):
* Primary Endpoint: Overall Response Rate (ORR) - the percentage of patients who had a complete or partial response to treatment.
* Secondary Endpoints:
* Progression-Free Survival (PFS) – time until disease gets worse or the patient dies.
* Time to Treatment Failure (TTF) – time until the patient stops treatment for any reason.
* Time to Next Therapy (TTNT) – time until the patient starts their next line of treatment.
* Overall Survival (OS) - time until death from any cause.
* additional (Not Reported in this Paper):
* Correlation between tumor tissue and circulating tumor DNA (ctDNA) molecular profiles.
* quality of life measurements.
* Immune fitness in both treatment groups.
* Association between molecular evaluation and gene expression profiling.
3. How Outcomes Were Assessed:
* Tumor Response: Evaluated using RECIST v1.1 and irRC criteria.Assessments continued even after treatment stopped (unless due to lack of consent) until disease progression was documented.
* Survival: Monitored until death, loss to follow-up, consent withdrawal, or study end.
* Adverse Events (AEs): Graded using CTCAE v5.0, and their frequency and severity were recorded.
4. Data Handling & Oversight:
* Data Collection: Collected at clinical sites and entered into an electronic case report form (eCRF).
* Autonomous Monitoring: An independent Data Monitoring Committee (DMC) oversaw the trial, reviewing safety and efficacy.
* Interim Analysis: A pre-planned analysis was conducted when 20% of patients (n=76) reached a specific time point. The DMC recommended continuing the trial based on a conditional power calculation (CPt) showing strong evidence of benefit with TT.
* Compliance: The trial was conducted according to the protocol, amendments, and Good Clinical practice guidelines.
5. Sample Size:
* Hypothesis: TT was expected to have a 20% ORR, while SoC was expected to have a 5% ORR.
* Cohorts: Patients were divided into four groups based on cancer type:
* Breast Cancer
* Non-Colorectal Gastrointestinal Cancers
* Non-Small Cell Lung Cancer (NSCLC)
* Othre Malignancies
* Total Sample Size: 86 patients (43 in each arm, across the four cohorts) were needed to detect a 15% difference in ORR with 80% power.
let me know if you’d like me to elaborate on any specific aspect of these methods!
