Guinea-Bissau Hepatitis B Vaccine Trial Criticised as Unethical | WHO
- A planned U.S.-funded trial to study the hepatitis B vaccine in newborns in Guinea-Bissau has been halted following criticism from the World Health Organization (WHO), which deemed the...
- The core of the WHO’s objection lies in the study’s design, which involved delaying vaccination for a portion of the newborns.
- The WHO’s statement, released on February 14, 2026, emphasized that trials involving placebos or delayed treatment are generally only justifiable when no established treatment exists.
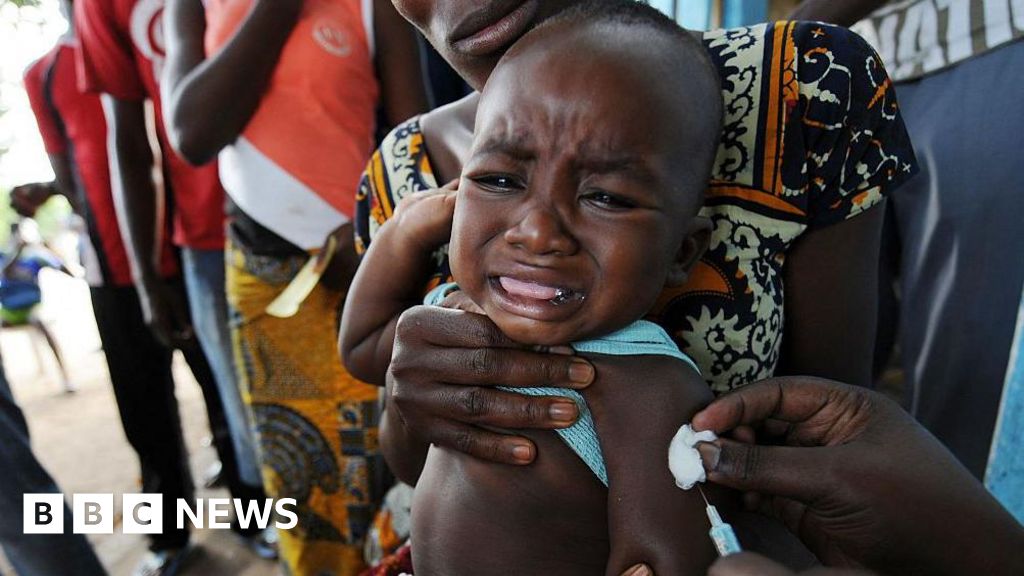
A planned U.S.-funded trial to study the hepatitis B vaccine in newborns in Guinea-Bissau has been halted following criticism from the World Health Organization (WHO), which deemed the study “unethical.” The trial, initially approved with $1.6 million in funding from the U.S. Centers for Disease Control and Prevention (CDC), aimed to compare the effects of administering the vaccine at birth versus six weeks of age.
Concerns Over Withholding a Proven Intervention
The core of the WHO’s objection lies in the study’s design, which involved delaying vaccination for a portion of the newborns. The WHO argues that withholding a proven, life-saving intervention – the hepatitis B birth dose vaccine – from some infants is unacceptable. According to the WHO, the vaccine is “an effective and essential public health intervention, with a proven record,” preventing transmission of the virus from mother to baby in 70-95% of cases.
The WHO’s statement, released on , emphasized that trials involving placebos or delayed treatment are generally only justifiable when no established treatment exists. This is not the case with the hepatitis B birth dose vaccine, which has been in use for over three decades in more than 115 countries.
Study Aims and Controversy
The U.S. Health department, currently headed by Robert F. Kennedy Jr., had expressed interest in using the trial to investigate broader health effects associated with the hepatitis B vaccine. The study intended to randomize 14,000 newborns to receive their first dose of the vaccine at either birth or six weeks, and then study differences in alleged safety outcomes.
This research plan has sparked considerable debate, with some experts drawing parallels to the infamous Tuskegee Experiment, a deeply unethical study conducted in the United States where African American men with syphilis were deliberately left untreated to study the disease’s natural progression. The comparison highlights concerns about exploiting a vulnerable population for research purposes.
Guinea-Bissau’s Response and Suspension
The Minister of Public Health in Guinea-Bissau, Quinhin Nantote, announced on , that the study was suspended pending review. Nantote, speaking in Portuguese through an interpreter, indicated a potential for complete cancellation of the trial. This decision followed mounting pressure from public health experts and the WHO’s strong condemnation.
Hepatitis B and the Importance of Early Vaccination
Hepatitis B is a viral infection that attacks the liver and can cause both acute and chronic disease. A significant portion of Guinea-Bissau’s population is estimated to have hepatitis B, making early intervention particularly crucial. Infection at birth is the most common pathway to chronic infection, with approximately 90% of newborns infected going on to become chronic carriers. Chronic hepatitis B can lead to serious complications, including cirrhosis, liver cancer, and liver failure.
The WHO recommends that all newborns receive the hepatitis B vaccine within 24 hours of birth. This early vaccination is a cornerstone of global efforts to control and eliminate hepatitis B transmission. The birth dose vaccine is particularly effective in preventing mother-to-child transmission, a major route of infection in areas with high prevalence rates.
Background on the Researchers and Funding
The research team leading the trial is headed by Christine Stabell Benn and her husband Peter Aaby of the Bandim Health Project, based at the University of Southern Denmark in Copenhagen. Benn and Aaby have faced previous criticism for their research related to vaccine safety. The CDC’s funding of the trial occurred after Kennedy, who has publicly questioned the effects of vaccines, appointed advisors to the agency who subsequently moved to abandon a long-standing universal recommendation for the hepatitis B birth dose vaccine.
Ethical Considerations in Clinical Trials
This case underscores the critical importance of ethical safeguards in clinical research, particularly when involving vulnerable populations. Key ethical principles, such as beneficence (maximizing benefits and minimizing harms), respect for persons (autonomy and informed consent), and justice (fair distribution of risks and benefits), must be rigorously upheld. The WHO’s concerns regarding the scientific justification, ethical safeguards, and consistency with established standards for research involving humans highlight the need for careful scrutiny of research protocols before implementation.
The suspension of this trial serves as a reminder of the ongoing debate surrounding vaccine research and the need for transparency, ethical oversight, and a commitment to protecting the health and well-being of all individuals, especially newborns.
