Immune System’s Role in Alzheimer’s and Parkinson’s Diseases
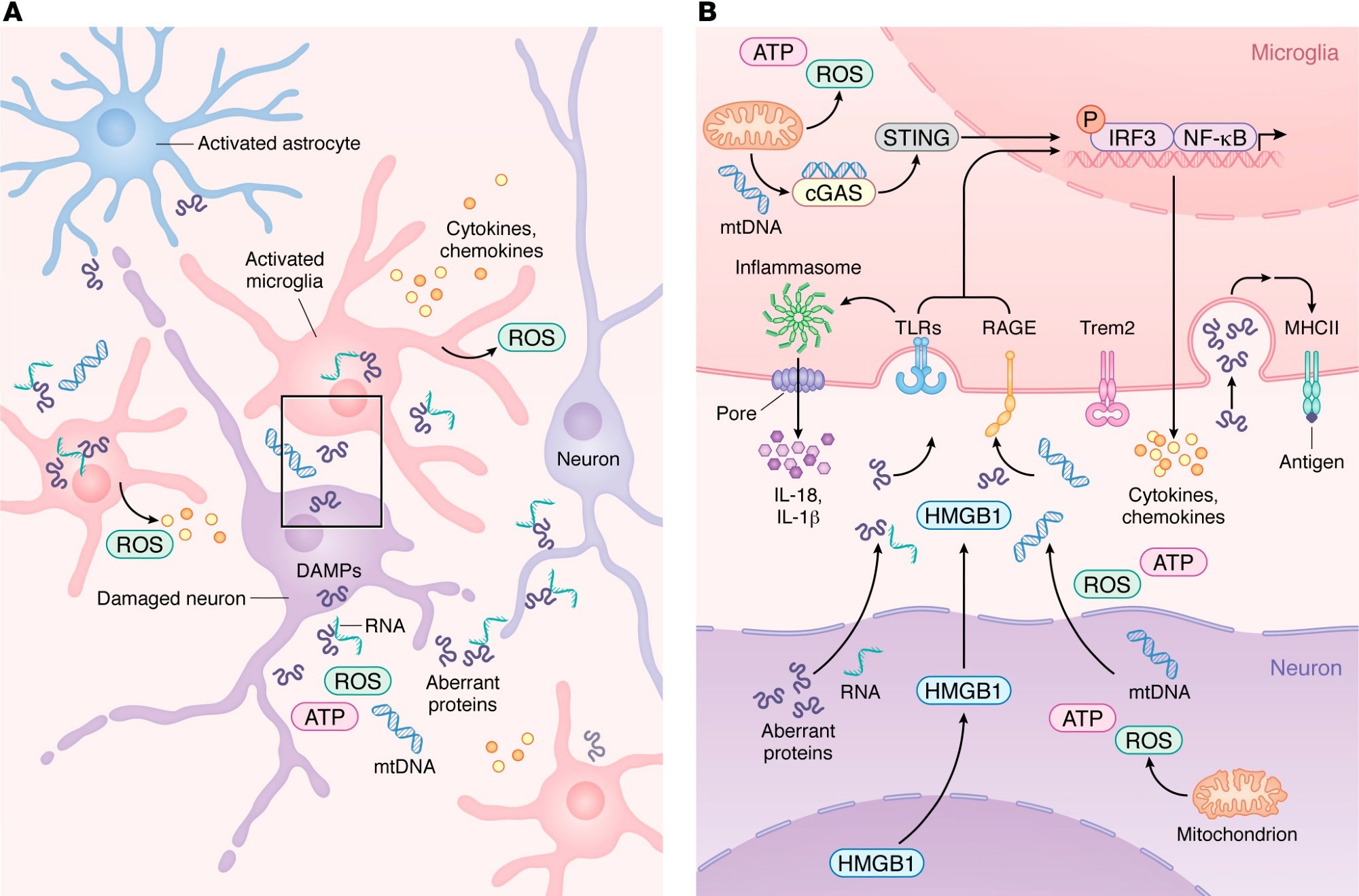
- The immune system plays a central role in the development and progression of neurodegenerative diseases such as Alzheimer’s and Parkinson’s, with growing evidence showing that chronic inflammation and...
- Research indicates that aging is the strongest risk factor for both Alzheimer’s disease and Parkinson’s disease and It's closely linked to changes in immune function that may promote...
- In Alzheimer’s disease, the accumulation of amyloid-beta plaques and tau tangles triggers activation of the brain’s resident immune cells, particularly microglia, which can become chronically activated and release...
The immune system plays a central role in the development and progression of neurodegenerative diseases such as Alzheimer’s and Parkinson’s, with growing evidence showing that chronic inflammation and immune dysregulation contribute significantly to neuronal damage.
Research indicates that aging is the strongest risk factor for both Alzheimer’s disease and Parkinson’s disease and It’s closely linked to changes in immune function that may promote neuroinflammation.
In Alzheimer’s disease, the accumulation of amyloid-beta plaques and tau tangles triggers activation of the brain’s resident immune cells, particularly microglia, which can become chronically activated and release inflammatory molecules that harm neurons.
Studies have also found that impairments in the blood-brain barrier allow peripheral immune cells, including B cells, to enter the central nervous system, where they may contribute to disease pathology through the production of autoantibodies and other immune mediators.
In Parkinson’s disease, alpha-synuclein aggregates activate microglial cells, leading to sustained inflammation and the release of cytotoxic substances that damage dopaminergic neurons in the substantia nigra.
Peripheral immune signaling molecules, such as cytokines, can influence brain function through the brain-peripheral immune axis, with models in organisms like Drosophila showing that pathways such as Toll, Imd, JAK/STAT, and MAPK are involved in regulating neuroimmune communication.
In these models, hyperactivation of the Imd pathway’s NF-κB homolog, Relish, in the central nervous system drives neurodegeneration through the neurotoxic effects of antimicrobial peptides, highlighting a conserved mechanism linking immune activation to neuronal loss.
These findings support the view that neurodegenerative diseases are not isolated to the brain but involve systemic immune dysregulation, suggesting that modulating immune responses could represent a therapeutic strategy.
However, researchers caution that the immune system has both protective and harmful roles in neurodegeneration, and timing, cell type specificity, and disease stage are critical factors that influence whether immune activity is beneficial or detrimental.
Ongoing research aims to clarify these dynamics to develop targeted immunotherapies that reduce harmful inflammation without compromising the immune system’s essential functions in brain surveillance, and repair.
