Integrating Animal, Human, and AI Models for Enhanced Translational Research
- Researchers should not abandon animal studies in favor of human-only or AI-based models but instead focus on integrating experimental systems to strengthen scientific confidence and translational relevance, according...
- The paper, titled “A roadmap to competitive preclinical packages,” argues that the ongoing debate over whether to replace animal research with human-derived or artificial intelligence models misses the...
- Elena Rodriguez, a pharmacologist at the Karolinska Institute in Sweden, emphasized that no single model currently captures the full complexity of human biology.
Researchers should not abandon animal studies in favor of human-only or AI-based models but instead focus on integrating experimental systems to strengthen scientific confidence and translational relevance, according to a new analysis published in Nature Medicine.
The paper, titled “A roadmap to competitive preclinical packages,” argues that the ongoing debate over whether to replace animal research with human-derived or artificial intelligence models misses the point. Rather than choosing one system over another, scientists should strategically combine multiple models — including animal, human tissue, organoid, and computational approaches — to maximize mechanistic understanding, human relevance, and real-world decision-making value in drug development.
Lead author Dr. Elena Rodriguez, a pharmacologist at the Karolinska Institute in Sweden, emphasized that no single model currently captures the full complexity of human biology. “Animal models remain indispensable for studying systemic physiology, long-term disease progression, and safety pharmacology,” she said. “But they have limitations in predicting human-specific responses, particularly in areas like immunology, and metabolism. The solution is not to discard them, but to use them alongside human-based systems where each compensates for the other’s weaknesses.”
The analysis highlights recent advances in human-relevant models, such as induced pluripotent stem cell (iPSC)-derived tissues, organ-on-a-chip platforms, and AI-driven predictive toxicology, which have improved the ability to study human-specific mechanisms. However, the authors note these tools often lack the integrated biological context — such as neuroendocrine feedback, immune trafficking, or multi-organ metabolism — that whole-animal studies provide.
To illustrate their framework, the researchers cite examples from cancer and metabolic disease research. In oncology, combining patient-derived xenografts in mice with genomic profiling of human tumors has helped identify biomarkers that predict response to targeted therapies. In diabetes research, studies using both rodent models and human islet transplants have clarified how beta-cell failure progresses under metabolic stress, leading to more accurate predictions of drug efficacy.
The authors propose a tiered approach to preclinical testing, where early mechanistic studies use human cell-based or computational models to screen candidates, followed by validation in animal models for systemic effects, and finally confirmation in human-relevant systems before clinical trials. This strategy, they argue, increases the likelihood of success in later stages while reducing reliance on any single model type.
Dr. James Liu, a bioengineer at the Massachusetts Institute of Technology not involved in the study, supported the call for integration. “The field is moving toward a ‘best evidence’ framework,” he said. “We’re not trying to eliminate animal use where it’s still scientifically necessary, but we are aiming to reduce unnecessary duplication and increase predictive power by using the right model for the right question.”
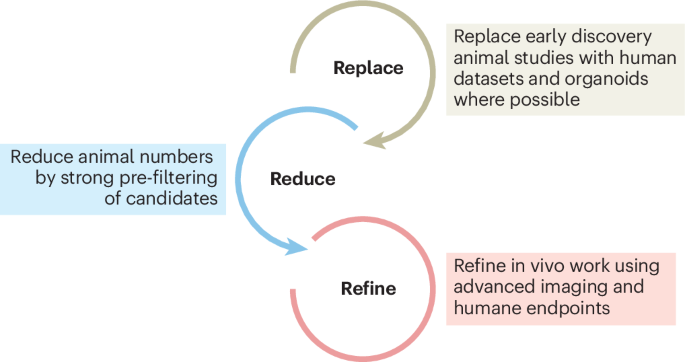
The paper acknowledges ethical and practical considerations in animal research, noting that efforts to refine, reduce, and replace animal use remain important. However, it cautions against framing the issue as a binary choice, which could lead to oversimplification and hinder scientific progress. Instead, the authors urge funding agencies, regulators, and pharmaceutical companies to invest in infrastructure that supports multi-model workflows and cross-platform data integration.
As drug development faces mounting pressures to improve efficiency and reduce failure rates — particularly in complex diseases like Alzheimer’s, heart failure, and autoimmune disorders — the authors conclude that a synergistic approach to preclinical modeling offers the most viable path forward. “The goal isn’t to pick a winner between species or systems,” Dr. Rodriguez said. “It’s to build a stronger, more reliable pipeline by letting each model do what it does best.”
