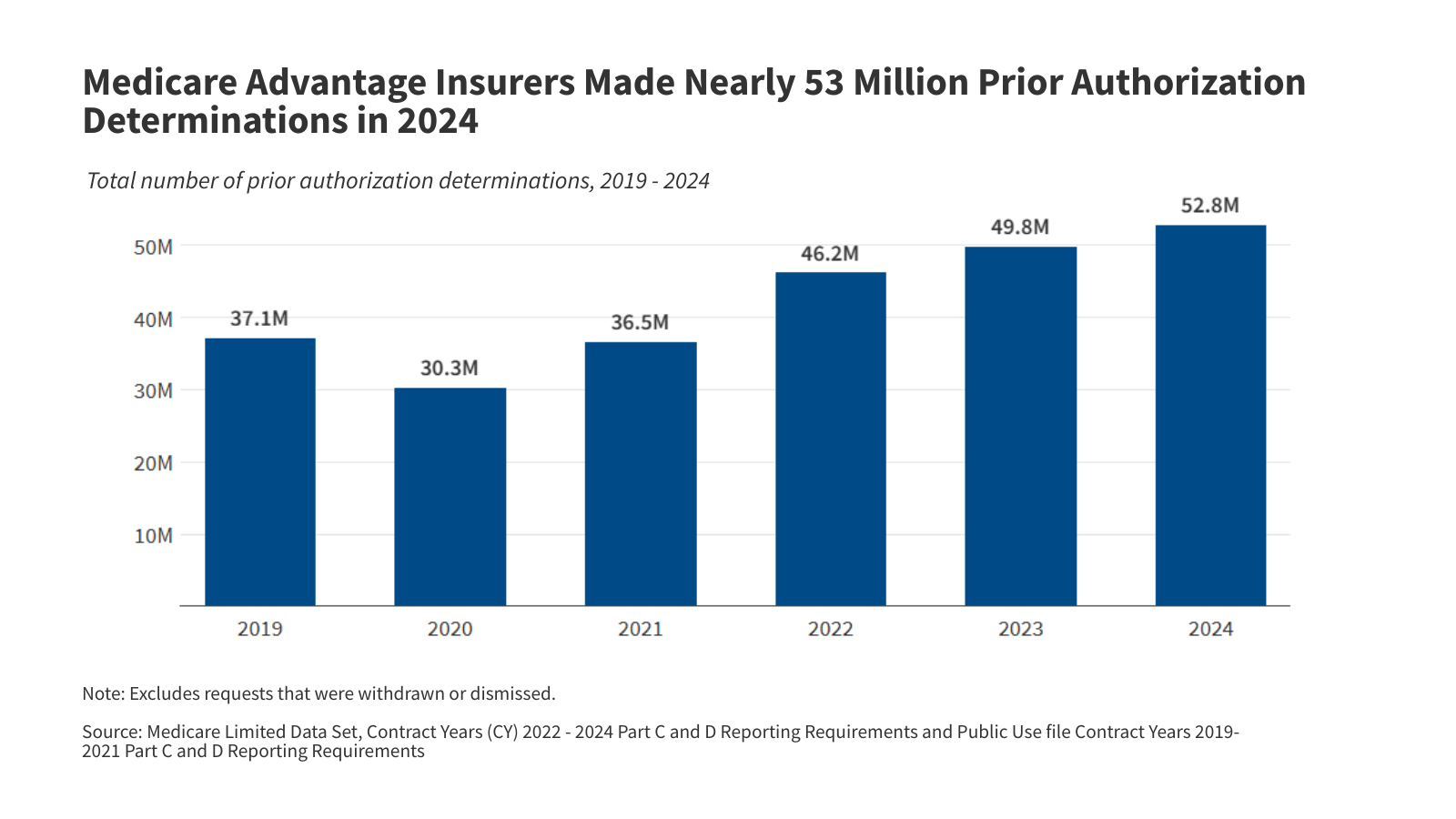
Medicare Advantage Prior Authorizations 2024
- As of January 28, 2026, Medicare requires prior authorization for over 70 Durable Medical Equipment, Prosthetics, Orthotics, and Supplies (DMEPOS) items to curb unnecessary utilization and associated costs.
- The Centers for Medicare & Medicaid Services (CMS) first established a prior authorization process for certain DMEPOS items wiht a final rule published on December 30,2015.
- according to CMS data, improper payments in the DMEPOS sector have historically been a significant concern.
Table of Contents
As of January 28, 2026, Medicare requires prior authorization for over 70 Durable Medical Equipment, Prosthetics, Orthotics, and Supplies (DMEPOS) items to curb unnecessary utilization and associated costs.
The Centers for Medicare & Medicaid Services (CMS) first established a prior authorization process for certain DMEPOS items wiht a final rule published on December 30,2015. This rule aimed to target items frequently billed in error or with questionable medical necessity. Implementation began on March 20, 2017, and the list of items requiring prior authorization has evolved thru subsequent rulemaking.
The initial focus was on reducing improper payments. according to CMS data, improper payments in the DMEPOS sector have historically been a significant concern. Prior authorization was seen as a key tool to address this issue.
For example, the Office of Inspector General (OIG) reports consistently highlight DMEPOS as an area prone to improper billing.
Currently, the list of DMEPOS items requiring prior authorization, updated January 13, 2026, includes more than 70 items. these include, but are not limited to:
- Pressure reducing support surfaces
- Power mobility devices (including complex power wheelchairs and scooters)
- Lower limb prosthetics
- continuous Glucose Monitors (CGMs) and related supplies
- Oxygen and oxygen equipment
This expansion demonstrates CMS’s ongoing effort to refine the program and address areas of high spending and potential fraud.
As an example, the inclusion of CGMs reflects the increasing cost of these devices and the need to ensure appropriate utilization based on medical necessity, as outlined in CMS Transmittal R3931CP.
Recent Updates: December 2025 Rulemaking
In December 2025, CMS issued a press release announcing changes to the prior authorization process. These changes aim to streamline the process for both providers and beneficiaries, and to improve the accuracy of prior authorization determinations.
Specifically, the final rule, detailed in the Federal Register, focuses on enhancing communication and transparency regarding prior authorization requests and appeals.
For instance,CMS is implementing a standardized prior authorization form and improving the electronic submission process,as stated in the CMS-1778-FC final rule.
