
New Drug Mimics High Altitude Health Benefits
Revolutionary Drug Mimics Mountain Air to Treat Rare Metabolic Diseases
Table of Contents
- Revolutionary Drug Mimics Mountain Air to Treat Rare Metabolic Diseases
- Revolutionary Drug Mimics Mountain Air to Treat Rare Metabolic Diseases
- Key Questions and Answers
- what is HypoxyStat and how does it work?
- How did HypoxyStat show promise in treating Leigh syndrome?
- What are the implications of HypoxyStat for the U.S. healthcare system?
- What are the future directions and challenges associated with HypoxyStat?
- What makes HypoxyStat a unique approach in the treatment of metabolic diseases?
- Conclusion
- Key Questions and Answers
by Newsdirectory3.com
Scientists are on the brink of a groundbreaking medical discovery with a new type of drug that mimics the physiological benefits of breathing in thin ‘mountain air’. This innovation could prove life-saving for individuals with serious metabolic diseases like Leigh syndrome, a rare and progressive condition that affects the body’s ability to use oxygen efficiently.
Continuous low-oxygen therapy provided by a daily pill could be a game-changer for patients with Leigh syndrome, a condition where mitochondria can’t use the body’s oxygen fast enough. Oxygen buildup in tissues can lead to damage and ultimately cell death. Children with Leigh syndrome often die within the first few years of life, with only about 20 percent making it to age 20.
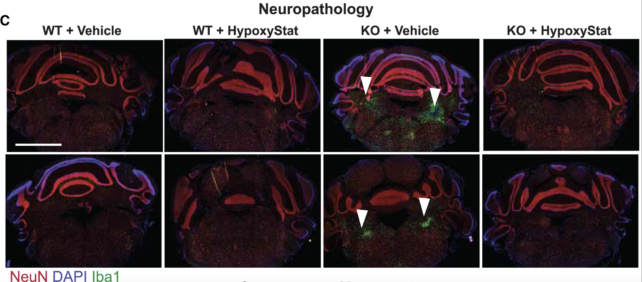
The new medicine, called HypoxyStat, extended the lifespan of mice with too much oxygen in their brains by up to 4-fold, reports a team from the Gladstone Institutes, the University of California San Francisco, and the pharmaceutical company Maze Therapeutics. In their proof-of-concept experiment, the drug worked even when given to mice in a late stage of neurodegeneration, where brain lesions are extensive and some animals are on the brink of death. The advanced damage was reversed, and movement, muscle weakness, and loss of coordination were improved.
Leigh syndrome is a rare, progressive disease where mitochondria can’t use the body’s oxygen fast enough. Oxygen buildup in tissues can lead to damage and ultimately cell death. Children with Leigh syndrome often die within the first few years of life. Only about 20 percent make it to age 20.
Typically, all but 2 percent of inhaled oxygen ends up in the body’s mitochondria – famously known as the ‘powerhouse’ of the cell because of its ability to use oxygen for energy production. But in high-altitude regions, the body shifts so that oxygen is not as easily delivered from the bloodstream to tissues.
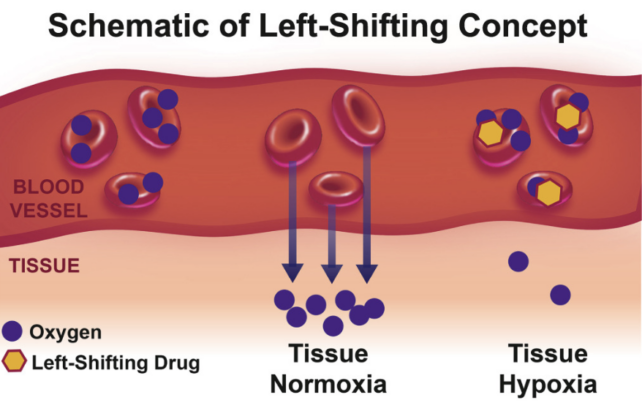
In a groundbreaking 2016 experiment, researchers found that for mouse models of Leigh syndrome, living in a low-oxygen environment not only prevents brain damage; it also reverses it. Further studies on mice in 2017 and 2019 confirmed those remarkable results.
It’s not clear if humans with Leigh syndrome respond in the same way to low-oxygen environments. But biochemist Isha Jain from the University of California San Francisco (UCSF) and her colleagues are already exploring a way to mimic those effects pharmaceutically.
Unlike sleeping in a hypobaric chamber, as some athletes do when training for high-altitude events, a drug that tricks the body into functioning like it would high up in the mountains could work virtually all day, leaving a person free to go about their usual business, inside or outside.
“It’s not practical for every patient with this disease to move to the mountains,” says Jain, an author on all three of the papers as well as the current study.
“But this drug could be a controlled and safe way to apply the same benefits to patients.”
Jain and her colleagues hypothesized that by making red blood cells carry more oxygen in the bloodstream, as they do in high-altitude environments, the amount of oxygen offloaded into the body’s tissues would be reduced.
In 2017, researchers found a drug, called GBT-440 or Voxelotor, that increases the affinity of the red cell protein hemoglobin to bind oxygen. When Jain and her team, led by biochemist Skyler Blume, incubated human red blood cells with GBT-440, they noticed the cells increased their affinity for hemoglobin by 75 percent.
Emboldened by their results, the researchers found another compound like GBT-440 and named it HypoxyStat.
Mice with Leigh syndrome that were treated with HypoxyStat showed a significant reduction in brain damage, shown by the green markers of lesions in the image below.
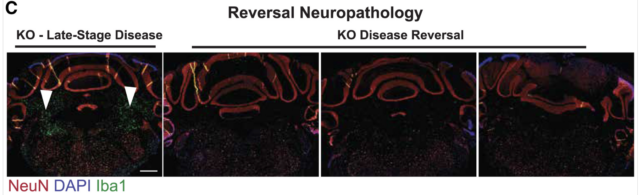
Even in later stages of the disease, a daily dose of this drug greatly extended mouse lifespans and reversed extensive damage in the brain.
“This ability to reverse advanced pathology positions HypoxyStat and related compounds as promising therapeutic candidates for mitochondrial diseases, where early diagnosis and intervention are often challenging,” the authors of the study conclude.
The research team is now exploring a second-generation version of HypoxyStat that could better translate to primate models or human clinical trials.
“Gas-based therapies for disease are really unique, and being able to bottle them up into drugs is a new, unusual concept,” says Jain.
“We’re excited to see where this promising strategy takes us.”
The study was published in Cell.
Implications for the U.S. Healthcare System
This breakthrough has significant implications for the U.S. healthcare system. Leigh syndrome and other mitochondrial diseases are often diagnosed late, making treatment challenging. HypoxyStat offers a new avenue for intervention, potentially improving the quality of life and lifespan for patients. The drug’s ability to reverse advanced pathology could be a game-changer, especially for conditions where early diagnosis is difficult.
Moreover, the potential for a daily pill to mimic the benefits of high-altitude living could revolutionize the treatment of various metabolic diseases. This approach could reduce the need for expensive and impractical interventions like moving to high-altitude regions or using hypobaric chambers.
The development of HypoxyStat also highlights the importance of interdisciplinary research. The collaboration between the Gladstone Institutes, the University of California San Francisco, and Maze Therapeutics exemplifies how combining expertise from different fields can lead to groundbreaking discoveries.
Future Directions and Challenges
The success of HypoxyStat in mice is a promising start, but translating this to human trials will be a significant challenge. Researchers will need to address potential side effects, optimize dosing, and ensure the drug’s efficacy in humans. Additionally, the regulatory process for new drugs is rigorous, requiring extensive clinical trials to prove safety and efficacy.
Despite these challenges, the potential benefits of HypoxyStat are immense. If successful, this drug could set a new standard for treating mitochondrial diseases and other conditions where oxygen metabolism plays a crucial role. The research team’s ongoing work on a second-generation version of the drug is a step in the right direction, aiming to better translate the findings to human trials.
Conclusion
The development of HypoxyStat represents a significant leap forward in the treatment of metabolic diseases. By mimicking the benefits of high-altitude living, this drug offers a novel approach to managing conditions like Leigh syndrome. As research continues, the potential for this therapy to improve the lives of patients is immense. The collaboration between leading institutions and pharmaceutical companies underscores the importance of interdisciplinary research in driving medical innovation.
Revolutionary Drug Mimics Mountain Air to Treat Rare Metabolic Diseases
Key Questions and Answers
what is HypoxyStat and how does it work?
Answer:
HypoxyStat is a groundbreaking drug that mimics the physiological benefits of breathing in thin ‘mountain air’. It targets conditions like Leigh syndrome, a rare metabolic disease where the body’s mitochondria cannot efficiently use oxygen, leading to oxygen buildup in tissues, damage, and ultimately cell death. By increasing the affinity of red blood cell hemoglobin for oxygen, HypoxyStat reduces oxygen delivered to tissues, akin to how bodies function in high-altitude environments.
- Key Insight: HypoxyStat functions by optimizing how red blood cells carry and deliver oxygen, decreasing tissue oxygenation and possibly reversing damage in diseases where normal oxygen delivery is impaired.
How did HypoxyStat show promise in treating Leigh syndrome?
Answer:
Animal studies have demonstrated notable results with HypoxyStat. In mice with Leigh syndrome, the drug extended lifespans by up to 4-fold and was effective even in late stages of neurodegeneration, reversing brain damage and improving muscle coordination and strength. These outcomes indicate HypoxyStat’s potential to reverse advanced disease conditions in mitochondrial disorders.
- Key Insight: HypoxyStat has shown the capacity to not only improve survival but also to reverse lesions and tissue damage in mouse models of Leigh syndrome, suggesting a revolutionary approach to treatment.
What are the implications of HypoxyStat for the U.S. healthcare system?
Answer:
HypoxyStat offers a promising new avenue for addressing the challenges of diagnosing and treating Leigh syndrome and other mitochondrial diseases. By potentially reversing advanced pathology, it could improve quality of life and extend lifespan for patients. Furthermore,this drug might reduce the need for impractical interventions like relocating to high-altitude areas,thus revolutionizing treatment options for various metabolic diseases.
- Key Insight: HypoxyStat could be a game-changer for the U.S. healthcare system, offering new opportunities for intervention in challenging-to-treat diseases and highlighting the importance of interdisciplinary research.
What are the future directions and challenges associated with HypoxyStat?
Answer:
While HypoxyStat has shown promising results in animal models, translating these findings to human trials poses significant challenges. The drug’s safety, dosage optimization, and efficacy in humans must be established through rigorous clinical trials. The research team is also developing a second-generation drug to improve translation to human trials. Despite these hurdles, the potential benefits of HypoxyStat for treating mitochondrial diseases are considerable.
- Key Insight: A triumphant transition from animal models to human clinical trials is crucial, requiring careful navigation of regulatory processes and ensuring HypoxyStat’s safety and efficacy.
What makes HypoxyStat a unique approach in the treatment of metabolic diseases?
Answer:
HypoxyStat stands out because it uses a novel pharmaceutical approach to mimic the beneficial physiological effects of high-altitude environments without the need for patients to actually reside at high altitudes or use hypobaric chambers. This method of ‘bottling up’ gas-based therapies into a drug form represents a unique and innovative concept in therapeutic strategies.
- Key Insight: The drug’s ability to offer continuous,low-oxygen therapy via a daily pill showcases an unconventional yet effective method that has significant implications for treating oxygen metabolism-related diseases.
Conclusion
Answer:
HypoxyStat represents a significant leap forward in metabolic disease treatment. By mimicking the high-altitude physiological effects, it offers a novel therapeutic approach that has the potential to improve patient outcomes dramatically. Continued interdisciplinary research and advancements in drug development will be essential for realizing HypoxyStat’s full potential in clinical practice.
- Key Insight: The collaboration between leading institutions and pharmaceutical companies underscores the critical role of interdisciplinary research in driving innovation and improving treatments for complex diseases.
By focusing on these key questions, the article provides a complete and engaging view of HypoxyStat’s potential, challenges, and implications, reinforcing its significance in the treatment of metabolic diseases.
