New Genetic Risk Factors for Atrial Fibrillation Identified
Genetic Discoveries Offer New Insights into Atrial Fibrillation
Table of Contents
- Genetic Discoveries Offer New Insights into Atrial Fibrillation
- Genetic Discoveries Offer New Insights into Atrial Fibrillation: Your Questions Answered
- What is Atrial Fibrillation (AFib)?
- How Common is AFib?
- What Are the Risks Associated with AFib?
- why is Understanding the Genetic Basis of AFib Vital?
- What Have Genome-Wide Association Studies (GWAS) Revealed About AFib?
- What New Genetic Discoveries Have Been Made?
- What are Genetic Loci?
- What is the Significance of the Expanded Genetic Map Findings?
- What is a Polygenic Risk Score (PRS)? How is it used in AFib?
- What Genes Were Identified in the Rare Genetic Variants Study?
- What Structural DNA Changes Were Linked to AFib Risk?
- How Does Deactivation of the KDM5B Gene Affect AFib?
- How Can These Genetic Discoveries Improve AFib Treatment?
- What are the Limitations in AFib Treatment?
- What Are the Future Research Directions?
- Summary of Key Findings:
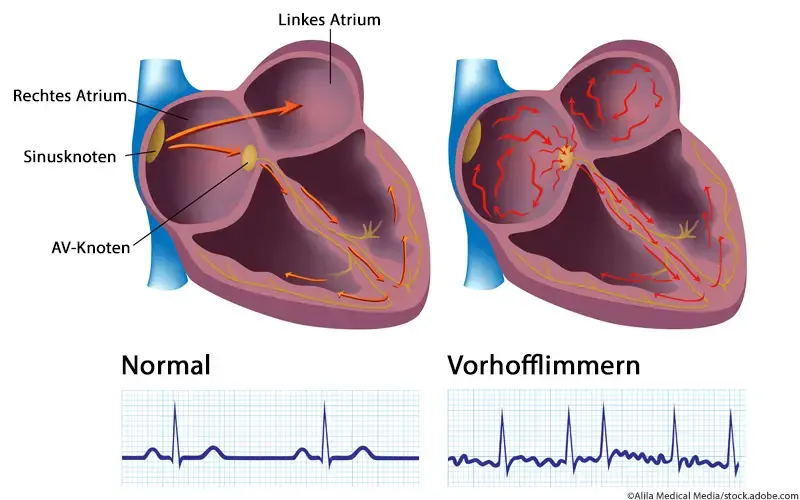
Atrial fibrillation (afib), the most prevalent sustained cardiac arrhythmia, affects millions globally. In Europe, it impacts roughly 2% to 3% of adults, with prevalence rising due to demographic shifts. The condition elevates the risk of stroke, heart failure, and mortality. Despite treatment advances, the underlying molecular mechanisms remain poorly understood, hindering the growth of targeted therapies.
Genome-wide association studies (GWAS) have previously linked numerous genetic variations to increased AFib risk.However, researchers suspected additional, undiscovered genetic factors were at play. A recent meta-analysis published in Nature Genetics expands this knowledge, providing deeper insights into the genetic architecture of AFib.
Expanded Genetic Map of Atrial Fibrillation
An international research team analyzed genetic data from over 180,000 AFib patients and nearly 1.5 million control subjects. By merging data from 68 studies worldwide, they identified over 350 genetic loci associated with increased AFib risk, doubling the number of previously known genetic risk regions.
Of particular significance is the discovery of 139 loci, including genes involved in muscle contractility, heart muscle development, and cell communication. These genes are highly active in atrial myocytes, suggesting a direct role in AFib pathogenesis.
The study also optimized risk prediction using a polygenic risk score (PRS). This PRS, based on the newly identified genetic variations, demonstrated superior predictive power for AFib risk compared to the previously used Charge AF Risk score. This advancement could improve the identification of high-risk patients and facilitate more targeted prevention strategies.
Rare Genetic Variants and Structural Heart Changes
A separate study, also published in nature genetics, analyzed rare genetic variants in over 50,000 AFib patients and more than 270,000 control subjects. Researchers identified new pathogenic variants in four genes: MYBPC3, LMNA, PKP2, and KDM5B. These genes are already implicated in cardiomyopathies, suggesting shared genetic underpinnings between structural heart disease and AFib.
Moreover, the research team identified structural DNA changes associated with increased AFib risk. Deletions in the CTNNA3 gene and duplications in the GATA4 gene appear to influence heart muscle structure and may contribute to atrial electrical instability.
an experimental approach using gene-editing techniques revealed that deactivation of the KDM5B gene in stem cell-derived atrial myocytes leads to alterations in electrical activity, a mechanism relevant to AFib.
Toward personalized prevention and Therapy
The findings from these two studies significantly enhance the understanding of the genetic basis of atrial fibrillation. Identifying new risk alleles could pave the way for personalized prevention and treatment strategies. The new polygenic risk score could be notably useful in identifying high-risk individuals, even those without symptoms, enabling early intervention.
“Atrial fibrillation is a common illness, but our pharmacological therapy options are still limited as we have so far insufficiently understand the underlying molecular mechanisms,” said Patrick Ellinor, director of the Cardiovascular Disease Initiative at the Broad Institute of MIT and Harvard. “These studies provide new potential goals for drug development.”
Future Research Directions
The newly identified genes and genetic variations require further investigation to elucidate their functional effects on AFib pathophysiology. Future research could determine whether targeted genetic interventions or personalized therapy approaches can reduce AFib risk or slow its progression.
“Large,international consortia such as the AFGen consortium play a crucial role in these investigations,” Ellinor stated. “No single study is large enough to deliver significant results. Through international cooperation and the open exchange of data, however, we can make progress that ultimately improve patient care.”
Genetic Discoveries Offer New Insights into Atrial Fibrillation: Your Questions Answered
Atrial fibrillation (AFib) is a common heart condition. Recent genetic studies have revealed new insights into its causes adn potential treatment pathways. Let’s dive into the details.
What is Atrial Fibrillation (AFib)?
Atrial fibrillation (AFib) is the most prevalent sustained cardiac arrhythmia, affecting millions globally. It’s a condition where the upper chambers of the heart (atria) beat irregularly and rapidly.
How Common is AFib?
in Europe, AFib impacts approximately 2% to 3% of adults, with prevalence increasing due to demographic shifts. The condition elevates the risk of stroke,heart failure,and mortality.
What Are the Risks Associated with AFib?
AFib significantly increases the risk of several serious health complications, including:
Stroke: Irregular heartbeats can cause blood clots, increasing the risk of stroke.
Heart Failure: The heart may not pump blood effectively, leading to heart failure.
Mortality: AFib can increase the risk of death.
why is Understanding the Genetic Basis of AFib Vital?
It is indeed critically important to understand the genetic basis of AFib as, despite treatment advances, the underlying molecular mechanisms remain poorly understood, hindering the growth of targeted therapies.
What Have Genome-Wide Association Studies (GWAS) Revealed About AFib?
Genome-wide association studies (GWAS) have previously linked numerous genetic variations to increased AFib risk.However, researchers suspected additional, undiscovered genetic factors were at play. A recent meta-analysis published in Nature Genetics expands this knowledge,providing deeper insights into the genetic architecture of AFib.
What New Genetic Discoveries Have Been Made?
Researchers have identified several new genetic factors related to AFib. These discoveries come from two primary research focuses, published in Nature Genetics:
Expanded Genetic Map: An international research team analyzed genetic data from over 180,000 AFib patients and nearly 1.5 million control subjects, identifying over 350 genetic loci associated with increased AFib risk.
Rare Genetic variants and Structural Heart Changes: A separate study analyzed rare genetic variants in over 50,000 AFib patients and more than 270,000 control subjects, identifying new pathogenic variants in four genes and structural DNA changes.
What are Genetic Loci?
Genetic loci are the specific locations of genes on a chromosome.
What is the Significance of the Expanded Genetic Map Findings?
The expanded genetic map identified over 350 genetic loci associated with increased AFib risk,including 139 loci with genes involved in muscle contractility,heart muscle growth,and cell communication. These genes are highly active in atrial myocytes, suggesting a direct role in AFib pathogenesis.
What is a Polygenic Risk Score (PRS)? How is it used in AFib?
A polygenic risk score (PRS) is a score based on multiple genetic variations to predict an individual’s risk for a particular disease.In these studies,a PRS was optimized using the newly identified genetic variations. This PRS demonstrated superior predictive power for AFib risk compared to the previously used Charge AF Risk score,which could help identify high-risk patients.
What Genes Were Identified in the Rare Genetic Variants Study?
The study identified new pathogenic variants in four genes:
MYBPC3
LMNA
PKP2
KDM5B
These genes are already implicated in cardiomyopathies (diseases of the heart muscle), suggesting shared genetic underpinnings between structural heart disease and AFib.
What Structural DNA Changes Were Linked to AFib Risk?
The research team found that structural DNA changes are associated with increased AFib risk. Specifically:
Deletions in the CTNNA3 gene.
Duplications in the GATA4 gene.
These changes may influence heart muscle structure and contribute to atrial electrical instability.
How Does Deactivation of the KDM5B Gene Affect AFib?
An experimental approach using gene-editing techniques revealed that deactivation of the KDM5B gene in stem cell-derived atrial myocytes leads to alterations in electrical activity,a mechanism relevant to AFib.
How Can These Genetic Discoveries Improve AFib Treatment?
Identifying new risk alleles could pave the way for personalized prevention and treatment strategies. The new polygenic risk score could be notably useful in identifying high-risk individuals, even those without symptoms, enabling early intervention.
What are the Limitations in AFib Treatment?
As stated by Patrick Ellinor, director of the Cardiovascular Disease Initiative at the Broad Institute of MIT and Harvard, “Atrial fibrillation is a common illness, but our pharmacological therapy options are still limited as we have so far insufficiently understand the underlying molecular mechanisms.”
What Are the Future Research Directions?
Future research will focus on:
Further investigation of the newly identified genes and genetic variations to understand their functional effects on AFib.
* Determining if targeted genetic interventions or personalized therapy approaches can reduce AFib risk or slow its progression.
Large international consortia, such as the AFGen consortium, play a crucial role in these investigations.
Summary of Key Findings:
Hear is a table summarizing the significant discoveries:
| Finding | Details | Potential Impact |
|---|---|---|
| Expanded Genetic Map | Identified over 350 genetic loci associated with increased AFib risk. | Provides new targets for drug development and personalized prevention strategies. |
| New Genes Identified | Pathogenic variants found in MYBPC3, LMNA, PKP2, and KDM5B. | Highlights shared genetic links between AFib and cardiomyopathies. |
| Structural DNA Changes | Deletions in CTNNA3 and duplications in GATA4. | Reveals impact on heart muscle structure and electrical stability. |
| Polygenic Risk Score (PRS) | Improved AFib risk prediction. | Could improve the identification of high-risk patients for early intervention. |
