Photoswitch Therapy Safe for Advanced Retinitis Pigmentosa: Phase 1 Trial Results
- Results from a first-in-human phase 1 clinical trial indicate that a novel photoswitch therapy can be safely administered to individuals with advanced retinitis pigmentosa (RP).
- Retinitis pigmentosa is a group of genetically heterogeneous inherited retinal diseases.
- The ABACUS-1 study was an open-label, dose-escalation trial designed to be gene-agnostic, meaning it was conducted regardless of the underlying genetic mutation causing the disease.
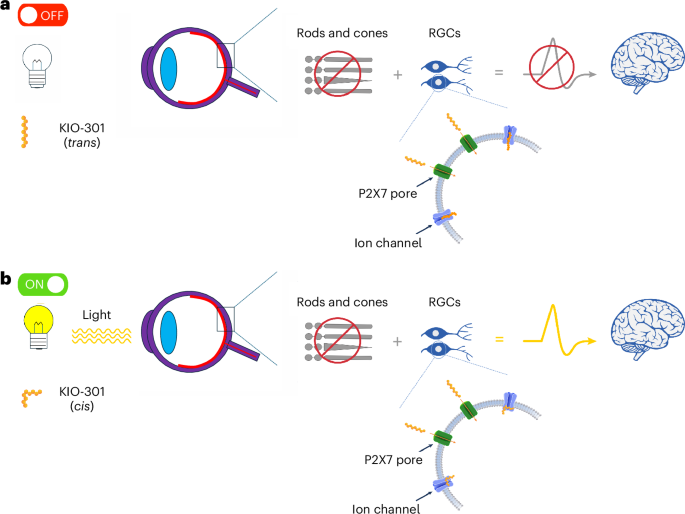
Results from a first-in-human phase 1 clinical trial indicate that a novel photoswitch therapy can be safely administered to individuals with advanced retinitis pigmentosa (RP). The study, published in Nature Medicine on April 14, 2026, evaluated the safety and feasibility of a small azobenzene photoswitch molecule known as KIO-301.
Retinitis pigmentosa is a group of genetically heterogeneous inherited retinal diseases. These conditions are characterized by the gradual degeneration of rod and cone photoreceptors, which typically leads to night blindness and a loss of the visual field, often progressing to profound visual impairment by middle age.
Trial Design and Primary Outcomes
The ABACUS-1 study was an open-label, dose-escalation trial designed to be gene-agnostic, meaning it was conducted regardless of the underlying genetic mutation causing the disease. KIO-301 was administered via intravitreal injection to 12 eyes across six participants with late-stage RP.

The primary objective of the trial was to assess ocular and systemic safety over a 30-day period. Researchers reported that the primary safety outcome was met, noting that no dose-limiting toxicities or serious adverse events occurred at any point during the study.
According to the findings, there was no drug-related intraocular inflammation. All observed ocular adverse events were classified as mild and related to the injection procedure rather than the drug itself.
Mechanisms and Exploratory Findings
KIO-301 is designed to confer light responsiveness to retinal ganglion cells, effectively bypassing the damaged photoreceptors in patients with advanced RP. To evaluate the efficacy of this mechanism, the researchers used several secondary and exploratory assessments, including functional vision testing, visual acuity and kinetic visual fields.
The trial utilized functional magnetic resonance imaging (fMRI) to monitor brain activity. The results showed light-evoked blood-oxygen-level-dependent signal changes in visual cortical regions following the dosing of KIO-301. These changes followed a temporal pattern that researchers stated was compatible with pharmacodynamic activity.
exploratory assessments revealed variation in light perception and functional vision measures among some of the participants. Participant-reported quality-of-life scores were also tracked, though these scores varied over time.
Clinical Implications and Future Research
While the phase 1 trial established an acceptable safety and tolerability profile, the investigators emphasized that these early results are not yet definitive regarding the restoration of daily vision.
The results of the trial provide evidence of short-term ocular safety and feasibility, while underscoring that larger, controlled studies are still needed to determine whether any functional changes translate into reliable, everyday vision benefit.
Dr. Robert Casson, Principal Investigator from the Royal Adelaide Hospital
Based on the data from the ABACUS-1 study, Kiora Pharmaceuticals has initiated a randomized, controlled Phase 2 clinical trial referred to as ABACUS-2. This subsequent study is intended to evaluate functional visual improvements and compare the results against a control group.
The transition to Phase 2 marks a shift from establishing safety to determining whether the molecular photoswitch can provide a consistent and meaningful improvement in the quality of vision for those living with late-stage inherited retinal diseases.
