Repotrectinib Shows Durable Response in NTRK Fusion–Positive Advanced Solid Tumors – TRIDENT-1 Trial
- A new study published today, February 4, 2026, in Nature Medicine offers encouraging long-term data on repotrectinib, a tyrosine kinase inhibitor showing promise for patients with advanced cancers...
- Repotrectinib works by selectively targeting ROS1, TRKA-C, and ALK – proteins that, when mutated, can fuel cancer growth.
- The TRIDENT-1 trial, an ongoing phase 1/2 study, has been evaluating repotrectinib in adult patients with advanced ROS1+ NSCLC or NTRK+ locally advanced or metastatic solid tumors.
A new study published today, , in Nature Medicine offers encouraging long-term data on repotrectinib, a tyrosine kinase inhibitor showing promise for patients with advanced cancers driven by specific genetic changes. The phase 1/2 TRIDENT-1 trial demonstrates that repotrectinib is safe and provides durable clinical responses, both within the tumor itself and in cases where cancer has spread to the brain, for individuals with NTRK fusion-positive advanced solid tumors.
Repotrectinib works by selectively targeting ROS1, TRKA-C, and ALK – proteins that, when mutated, can fuel cancer growth. This makes it a potentially valuable treatment option for cancers harboring these specific genetic fusions, regardless of where the cancer originates in the body. The treatment is already approved for adult patients with locally advanced or metastatic ROS1 fusion-positive non-small cell lung cancer (NSCLC), and for adult or pediatric patients aged 12 years or younger with locally advanced or metastatic NTRK fusion-positive solid tumors.
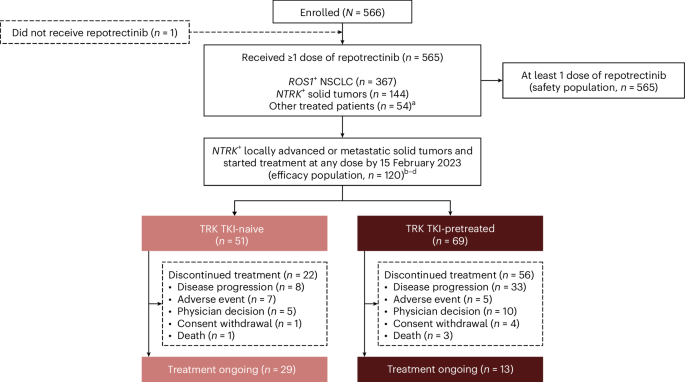
The TRIDENT-1 trial, an ongoing phase 1/2 study, has been evaluating repotrectinib in adult patients with advanced ROS1+ NSCLC or NTRK+ locally advanced or metastatic solid tumors. The study divided participants in the initial phase into primary and expanded efficacy populations, both receiving doses ranging from 40 to 160 mg of repotrectinib once daily. The primary efficacy population included patients who began treatment before , while the expanded efficacy population included those who started treatment before .
Researchers also tracked a safety population receiving the recommended phase 2 dose of repotrectinib. The study included patients who had not previously received tyrosine kinase inhibitors (TKI-naïve, n=71) and those who had (TKI-pretreated, n=56). Median follow-up was substantial, at 44.6 months for TKI-naïve patients and 42 months for those previously treated with TKIs, in the primary efficacy cohort. The expanded efficacy cohort saw median follow-up periods of 37.7 and 34.8 months, respectively.
The data presented at JADPRO Live 2025, in National Harbor, Maryland, by Misako Nagasaka, MD, PhD, of the University of California Irvine, and led by principal investigator Byoung Chul Cho, MD, PhD, of the Yonsei Cancer Center, highlight the durability of response seen with repotrectinib. This is particularly important as patients with NTRK-positive cancers often face significant unmet needs, and the development of treatments that can provide lasting benefits is crucial.
In , the Food and Drug Administration (FDA) granted priority review to a supplemental new drug application (sNDA) for repotrectinib as a treatment for patients 12 years and older with locally advanced or metastatic solid tumors unsuitable for surgical resection and harboring an NTRK gene fusion. The FDA has set a Prescription Drug User Fee Act (PDUFA) date of , for its decision on the application.
According to Bristol Myers Squibb, the developer of repotrectinib, the priority review was supported by data from the TRIDENT-1 trial (NCT03093116). Joseph Fiore, vice president and global program lead of repotrectinib at Bristol Myers Squibb, stated that new and effective treatment options are critical for patients with NTRK-positive locally advanced or metastatic solid tumors, and that repotrectinib may offer a durable treatment option.
The study also examined patients with measurable brain metastases at the start of treatment, suggesting that repotrectinib can effectively target cancer that has spread to the brain. Updated data from the trial, presented in , continued to demonstrate high response rates and durable responses, including robust intracranial responses.
Findings published in Annals of Oncology showed a confirmed objective response rate with repotrectinib in patients with NTRK-positive tumors. While specific response rates were not detailed in the available information, the consistent reporting of durable responses across multiple data releases suggests a clinically meaningful benefit for patients.
Repotrectinib’s approval pathway and ongoing research underscore the growing importance of precision medicine in cancer treatment. By targeting specific genetic drivers of cancer, therapies like repotrectinib offer the potential for more effective and personalized treatment strategies. The TRIDENT-1 trial continues to provide valuable insights into the long-term impact of this therapy and its potential to improve outcomes for patients with NTRK fusion-positive cancers.
