Revolutionizing Green Hydrogen Production: New Chiral Catalyst Speeds Up Water Splitting
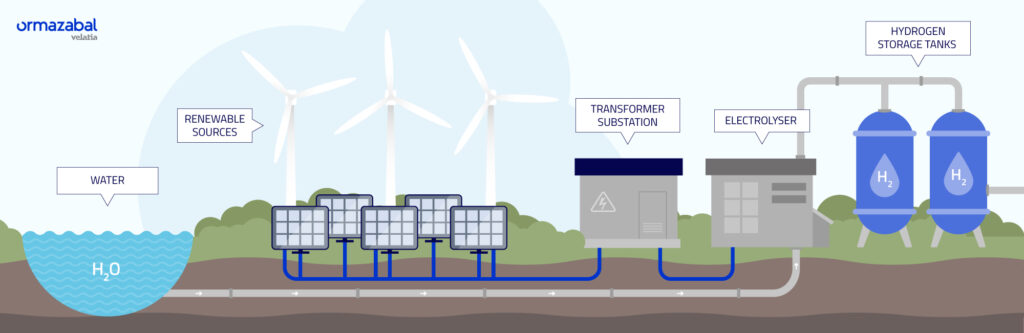
- Hydrogen is gaining importance in the transition to cleaner energy.
- A recent study in Nature Energy introduced a new method for producing hydrogen through water splitting.
- The new technique uses special crystals with chiral structures.
Hydrogen is gaining importance in the transition to cleaner energy. It is versatile and can serve as fuel, raw material for industries, or energy storage. When produced sustainably, hydrogen provides a clean alternative to fossil fuels, helping reduce greenhouse gas emissions and fight climate change.
A recent study in Nature Energy introduced a new method for producing hydrogen through water splitting. This process separates water into hydrogen and oxygen. The study focuses on producing “green hydrogen,” which occurs without fossil fuels.
The new technique uses special crystals with chiral structures. These crystals have left or right-handed atomic arrangements that improve the water-splitting process. They consist of rhodium and elements like silicon, tin, and bismuth. These components enhance electron transfer efficiency, speeding up the chemical reaction.
Dr. Xia Wang, a lead researcher from the Max Planck Institute, stated, “These crystals are essentially quantum machines. By leveraging the unique spin properties of electrons, we’ve created a catalyst that outperforms traditional materials by a factor of 200.”
Water splitting is often slow due to the oxygen evolution reaction (OER), which is inefficient. Chiral catalysts improve performance because they remain stable under tough conditions and do not require costly materials like platinum. This research could lead to better, cheaper catalysts for water splitting.
The study was conducted by scholars from the Max Planck Institute and the Weizmann Institute of Science. They explored this area to advance renewable energy technology and improve hydrogen production methods.
