Risk Assessment in Sterile Pharmaceutical Quality Assurance
- Risk assessment is being utilized as a process quality assurance tool to enhance contamination control within sterile pharmaceutical manufacturing.
- This approach falls under Quality Risk Management (QRM), which is identified as a risk-based approach to quality management.
- The systematic process of Quality Risk Management involves several key stages to manage risks facing the quality of pharmaceutical products throughout their development.
Risk assessment is being utilized as a process quality assurance tool to enhance contamination control within sterile pharmaceutical manufacturing. According to News-Medical, applying these risk assessment processes improves the overall quality assurance of manufacturing operations.
This approach falls under Quality Risk Management (QRM), which is identified as a risk-based approach to quality management. As detailed by MDPI, QRM is a systematic process used to ensure that pharmaceutical products remain safe and effective by proactively identifying potential risks.
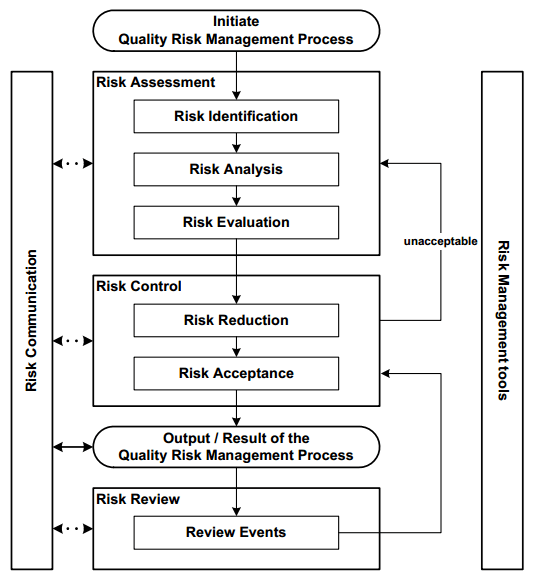
The Quality Risk Management Process
The systematic process of Quality Risk Management involves several key stages to manage risks facing the quality of pharmaceutical products throughout their development. These stages include:
- Assessment of risks
- Control of risks
- Communication regarding risks
- Review of risks
By implementing these steps, manufacturers can address potential quality issues before they affect the final pharmaceutical product.
Contamination Control in Sterile Manufacturing
In the production of sterile pharmaceuticals, environmental contamination control is a critical component. Risk management is necessary to ensure that these controls are effective and that the sterile environment is maintained.
The application of risk assessment tools allows manufacturers to better manage the variables that could lead to contamination, thereby safeguarding the integrity of the sterile manufacturing process.
Regulatory Context
The implementation of these quality assurance tools occurs as the regulatory environment for pharmaceutical products evolves. Contract Pharma reports that the regulatory bar is changing specifically for sterile injectables, necessitating rigorous quality management and risk-based approaches.
