SETDB1 Regulates Neuroinflammation via Neuronal P2rx7 Expression
- Research published in Nature has identified a specific epigenetic mechanism that regulates neuroinflammation in the mouse cortex, centering on the histone H3K9me3 methyltransferase SETDB1 and its interaction with...
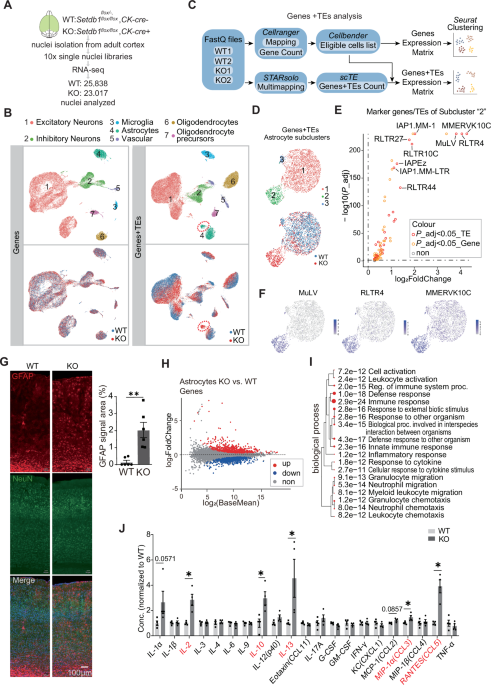
- The study demonstrates that the loss of SETDB1 in excitatory neurons leads to the activation of endogenous retroviruses (ERVs) and subsequent neuroinflammation.
- Researchers identified the P2rx7 gene as a direct target of SETDB1.
Research published in Nature has identified a specific epigenetic mechanism that regulates neuroinflammation in the mouse cortex, centering on the histone H3K9me3 methyltransferase SETDB1 and its interaction with the purinergic receptor gene P2rx7. The findings suggest a potential pathway for addressing neuroinflammatory responses associated with neuropsychiatric conditions.
The study demonstrates that the loss of SETDB1 in excitatory neurons leads to the activation of endogenous retroviruses (ERVs) and subsequent neuroinflammation. This process occurs in both neurons and astrocytes, resulting in the development of a reactive astrocyte population characterized by an increase in inflammatory cytokines and chemokines.
The Role of P2rx7 and Epigenetic Silencing
Researchers identified the P2rx7 gene as a direct target of SETDB1. The study characterized a novel enhancer located in the first intron of P2rx7 that is epigenetically silenced by SETDB1. When SETDB1 is absent, there is an increase in chromatin accessibility, which leads to the aberrant overexpression of P2rx7.
This specific epigenetic regulatory mechanism is reported to be conserved between humans and mice. In experimental models, the genetic ablation of P2rx7 in mice lacking SETDB1 partially reversed the activation of ERVs and the dysregulation of inflammatory genes, while also attenuating synaptic dysfunction.
Broader Implications for Neuroinflammation
SETDB1 is known to catalyze the di- and tri-methylation of H3K9 to repress gene expression and is involved in neuronal differentiation and neurodevelopment. The ability of SETDB1 to modulate the immune response in the central nervous system provides a mechanistic basis for understanding how epigenetic changes initiate neuroinflammatory responses.
Separate research into obstructive sleep apnea (OSA) has further highlighted the role of SETDB1 in microglia-mediated neuroinflammation. In studies involving C57BL/6J mice exposed to intermittent hypoxia for four weeks, researchers observed substantial increases in SETDB1 and H3K9me3 levels both in vivo and in vitro.
In the context of OSA, the upregulation of SETDB1 was found to increase the transcription factors p-NF-κB and p-signal transducer and activator of transcription 3 (p-STAT3). The inhibition of SETDB1 in microglia, whether through pharmacological or genetic means, resulted in a decrease in the induction of proinflammatory factors and a reduction in the generation of reactive oxygen species (ROS).
Potential Therapeutic Interventions
The research into OSA-induced cognitive impairment explored the use of acetate (ACE) to mitigate these effects. Findings indicated that supplementation with glyceryl triacetate (GTA) significantly mitigated cognitive deficits in mice exposed to intermittent hypoxia.
The mechanistic data suggests that acetate supplementation effectively represses high levels of SETDB1 and H3K9me3, which in turn inhibits the pro-inflammatory responses of microglia. In vivo, this supplementation reduced hippocampal levels of pro-inflammatory cytokines and p-NF-κB and p-STAT3, while protecting neuronal integrity.
Together, these studies point toward SETDB1 and its downstream targets, such as P2rx7, as significant targets for pharmacological intervention in treating neuroinflammatory conditions across different triggers, from genetic loss to environmental stressors like intermittent hypoxia.
