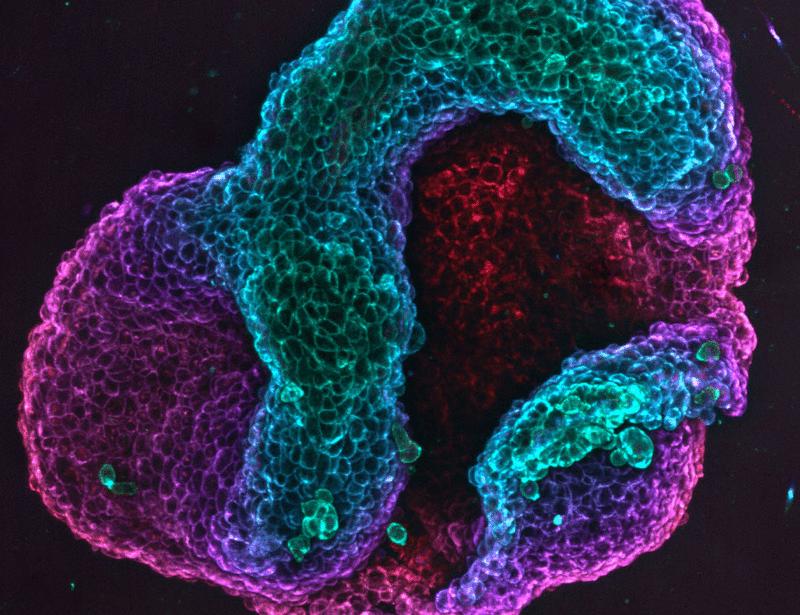
Topological Defects Shape Living Cell Sheets Into Predictable Structures
- Researchers have discovered that topological defects in living cell sheets can encode geometric features that guide the predictable folding of tissues into specific three-dimensional shapes, such as bowls...
- Dunlop and Łucja Kowalewska, demonstrates that these defects—disruptions in the otherwise orderly packing of cells—are not merely errors but carry critical positional information.
- Topological defects arise when cells in a sheet cannot align perfectly due to geometric constraints, creating points where the orientation of cellular structures—such as cytoskeletal filaments or cell...
Researchers have discovered that topological defects in living cell sheets can encode geometric features that guide the predictable folding of tissues into specific three-dimensional shapes, such as bowls and roses. This finding, published in the journal Science on April 16, 2026, reveals how inherent irregularities in cellular arrangements act as blueprints for morphogenesis, offering new insights into how complex biological structures form during development and regeneration.
The study, led by John W. C. Dunlop and Łucja Kowalewska, demonstrates that these defects—disruptions in the otherwise orderly packing of cells—are not merely errors but carry critical positional information. By analyzing patterns in active cell monolayers, the team showed that the type and arrangement of defects determine how a flat sheet of cells will bend, curve, or fold over time, effectively programming its final shape without external molds or forces.
How Defects Guide Tissue Folding
Topological defects arise when cells in a sheet cannot align perfectly due to geometric constraints, creating points where the orientation of cellular structures—such as cytoskeletal filaments or cell polarity—changes abruptly. These points are classified by their winding number, a mathematical property that remains stable unless the tissue undergoes significant reorganization. The researchers found that defects with integer winding values, such as +1 or -1, serve as stable markers that influence local tissue curvature and directional growth.

Programmable Morphogenesis in Cell Sheets
In laboratory experiments, the team began with flat, genetically identical sheets of epithelial cells and observed how they spontaneously folded into complex shapes over several days. By manipulating the initial density and alignment of cells, they could control the number and placement of topological defects. Sheets with a defined pattern of +1 defects folded upward into bowl-like structures, while alternating arrangements of +1 and -1 defects produced saddle-like or rose-like configurations.
The predictability of these outcomes suggests that topological defect patterns function as a kind of genetic or biophysical code. Rather than relying solely on biochemical gradients or mechanical tugs from surrounding tissues, the intrinsic organization of the cell sheet itself can direct morphogenesis through self-organizing principles rooted in physics and geometry.
Implications for Developmental Biology and Regenerative Medicine
This mechanism may help explain how embryos generate complex folded structures—such as the neural tube, gut, or lungs—without precise external instruction. If topological defects serve as evolutionarily conserved cues for tissue folding, they could represent a fundamental layer of biological organization that operates alongside genetic and chemical signaling pathways.
In regenerative medicine, the ability to predict and control tissue shape through defect engineering could improve the design of lab-grown tissues and organs. By introducing specific defect patterns into stem cell-derived sheets, scientists might guide the formation of functional vascular networks, bronchial trees, or intestinal villi without relying on complex scaffolding or external stimulation.
The researchers caution that while the principles are robust in simplified model systems, applying them to more complex, in vivo environments will require further study. Factors such as cell proliferation, apoptosis, and dynamic tissue adhesion may influence defect stability and behavior in developing embryos or healing wounds.
Future work will focus on identifying the molecular mechanisms that link defect signals to cellular behavior, such as changes in gene expression, adhesion, or cytoskeletal dynamics. The team also aims to explore whether similar principles apply to other types of tissues, including mesenchymal or neuronal cultures, and whether defect patterns can be edited using optogenetic or pharmacological tools to correct developmental abnormalities.
