Trabectedin and Irinotecan Combination Shows Promise for Ewing Sarcoma
- In a phase 1/2 clinical trial published in Nature Medicine on April 16, 2026, researchers found that combining trabectedin with low-dose irinotecan showed encouraging clinical activity in patients...
- The study, known as SARC037, enrolled 37 patients whose tumors had progressed despite prior treatments and who had no known effective therapy options.
- Trabectedin was administered as a one-hour intravenous infusion at a dose of 1.0 mg/m² on day 1 of each 21-day cycle, combined with low-dose irinotecan at 25 mg/m²...
In a phase 1/2 clinical trial published in Nature Medicine on April 16, 2026, researchers found that combining trabectedin with low-dose irinotecan showed encouraging clinical activity in patients with relapsed or refractory Ewing sarcoma, a cancer driven by the EWS::FLI1 transcription factor.
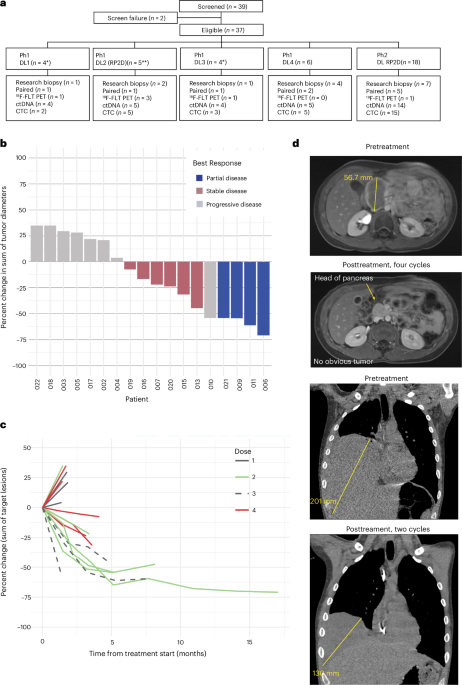
The study, known as SARC037, enrolled 37 patients whose tumors had progressed despite prior treatments and who had no known effective therapy options. All participants had measurable disease and an EWS-FLI1 fusion, the genetic abnormality that defines Ewing sarcoma and is essential for tumor cell survival.
Trabectedin was administered as a one-hour intravenous infusion at a dose of 1.0 mg/m² on day 1 of each 21-day cycle, combined with low-dose irinotecan at 25 mg/m² given on days 2 and 4. This regimen was selected based on preclinical evidence showing that trabectedin, at sufficient concentrations, can reverse the activity of EWS::FLI1, and that low-dose irinotecan enhances this effect.
The primary goals of the phase 2 portion of the trial were to assess safety, tolerability, and objective response rate (ORR). The recommended phase 2 dose (RP2D) was established during the phase 1 component as trabectedin 1.0 mg/m² over one hour and irinotecan 25 mg/m² on days 2 and 4.
At the RP2D, the most common severe side effects (grade 3 or higher) were myelosuppression and elevated alanine aminotransferase levels, each occurring in more than 15% of patients. These toxicities were described as manageable by the research team.
The objective response rate in the phase 2 portion of the trial was 33%. When including patients treated at the RP2D during the phase 1 stage, the ORR increased to 39%. The six-month progression-free survival rate was 48%.
To better understand the drug’s mechanism, researchers performed transcriptional profiling on tumor samples from a subset of patients. This analysis revealed a reversal of the EWS::FLI1-driven gene expression pattern in some tumors, indicating that the treatment successfully disrupted the core oncogenic driver in those cases.
Correlative studies in the trial also included molecular profiling, circulating tumor DNA analysis, pharmacokinetic evaluations, and imaging with 18F-fluorothymidine positron emission tomography (18F-FLT PET) to assess tumor proliferation changes during treatment.
The findings from SARC037 provide a foundation for further development of trabectedin combined with irinotecan as a targeted approach for Ewing sarcoma. The researchers emphasized that these results support continued investigation by international cooperative groups to refine treatment strategies for this aggressive malignancy.
