Wearable Optical Sensor Detects Glucose in Sweat for Diabetes Monitoring
- Continuous glucose monitoring is a cornerstone of diabetes management, but current methods often involve invasive sensors that can cause discomfort and potential complications.
- Diabetes requires frequent monitoring to prevent serious health consequences.
- The newly developed system addresses these challenges through a combination of nanostructured plasmonic materials and molecular recognition chemistry.
Continuous glucose monitoring is a cornerstone of diabetes management, but current methods often involve invasive sensors that can cause discomfort and potential complications. Researchers are now reporting a significant step forward: a portable, non-invasive system capable of detecting glucose levels directly from human sweat. This innovation, detailed in a study published on January 26, 2026, in Microsystems &. Nanoengineering, offers a promising alternative for the millions worldwide living with diabetes.
Diabetes requires frequent monitoring to prevent serious health consequences. Traditional methods rely on subcutaneous electrochemical sensors, which, while effective, carry risks of infection, inflammation, and reduced patient compliance over time. The appeal of sweat as a biofluid for glucose monitoring lies in its non-invasive accessibility and its correlation with blood glucose levels. However, sweat glucose concentrations are significantly lower—10 to 100 times lower—than those found in blood, and can be obscured by other compounds present in sweat, presenting a significant analytical challenge.
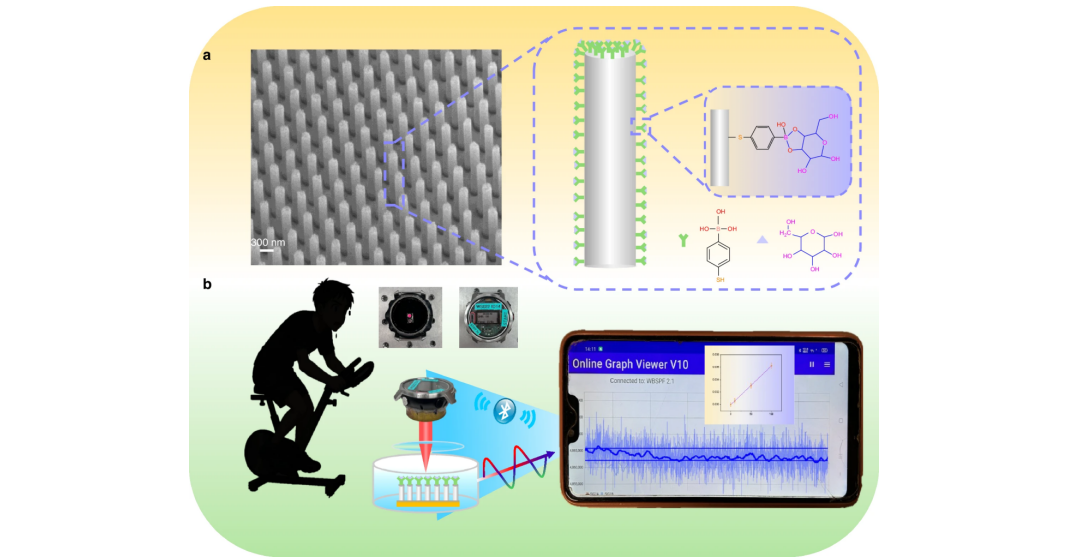
The newly developed system addresses these challenges through a combination of nanostructured plasmonic materials and molecular recognition chemistry. At its core is a silicon nanopillar array coated with silver. These nanopillars are engineered to generate a strong localized surface plasmon resonance when exposed to visible light. Crucially, the nanopillars are functionalized with 4-mercaptophenylboronic acid (4-MPBA), a molecule that selectively binds to glucose. This binding alters the optical properties of the nanopillar, creating a measurable signal without the need for enzymes or fluorescent labels.
The researchers optimized the system using both Raman spectroscopy and plasmonic reflectance measurements, demonstrating reliable glucose detection across a range of physiologically relevant concentrations. By utilizing silver instead of the more commonly used gold coating, they achieved sharper plasmonic responses and a detection limit as low as approximately 22 μmol/L – a level well within the range of glucose concentrations typically found in human sweat.
To translate this laboratory success into a practical, wearable device, the team integrated the sensor into an optical watch prototype. This prototype includes a compact LED light source, a photodiode to detect changes in light intensity, and a Bluetooth module for wireless data transmission to a smartphone application. When tested with both artificial sweat and samples collected from human volunteers during exercise, the system successfully tracked sweat glucose levels in real-time, demonstrating good agreement with standard enzymatic assays.
“Non-invasive glucose monitoring has long been limited by sensitivity and system complexity,” explained a senior researcher involved in the study. “By combining plasmonic nanostructures with a simple optical readout, we were able to detect glucose in sweat using low-power visible light. Importantly, this approach avoids enzymes and invasive probes, which opens new possibilities for comfortable, long-term monitoring. Our results show that wearable photonic sensors can move beyond the laboratory and into everyday health applications.”
This technology has the potential to significantly improve the quality of life for individuals who require frequent glucose monitoring, reducing discomfort, skin irritation, and the ongoing maintenance associated with traditional methods. Beyond diabetes, the modular design of the system allows for adaptation to detect other biomarkers present in sweat, such as lactate, electrolytes, or metabolites associated with stress. Future development could include automated sweat stimulation and microfluidic sampling to create a fully autonomous “lab-on-a-watch” device.
The research, supported by the Tandem Industry Academia 2021 project (No. 312) and the DigiHealth project (No. 326291) at the University of Oulu, highlights the convergence of nanophotonics and wearable electronics in enabling personalized, real-time health monitoring. While further clinical validation is necessary, this innovation represents a promising step towards a future where continuous health monitoring is seamless, comfortable, and readily accessible.
