White Matter Damage Drives Grey Matter Inflammation, Synapse Loss, and Neurodegenerative Progression
- A new study published in Nature reveals that damage to the brain’s white matter can trigger a cascade of events leading to grey matter inflammation and synapse loss,...
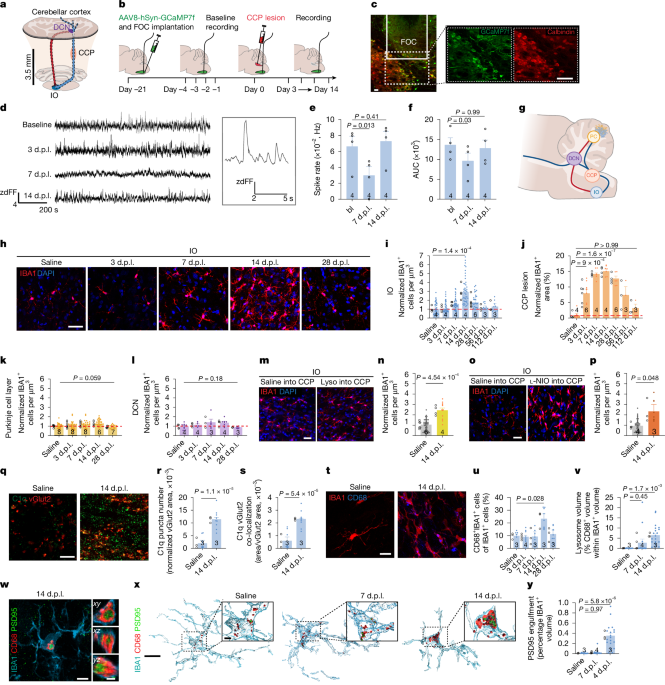
- Researchers created focal white matter lesions in rat brains within clinically relevant neural circuits to observe their effects.
- Grey matter microgliosis is often considered to be detrimental; however, we show that it is an integral part of regeneration and is conserved across three distinct mouse circuits...
A new study published in Nature reveals that damage to the brain’s white matter can trigger a cascade of events leading to grey matter inflammation and synapse loss, challenging long-held assumptions about the origins of neurodegenerative diseases like Alzheimer’s and Parkinson’s.
Researchers created focal white matter lesions in rat brains within clinically relevant neural circuits to observe their effects. They found that these lesions initially cause transient changes in neuronal activity and activate microglia in the grey matter, followed by synapse loss. Importantly, this grey matter response is not merely a consequence of damage but plays an active role in myelin regeneration.
Grey matter microgliosis is often considered to be detrimental; however, we show that it is an integral part of regeneration and is conserved across three distinct mouse circuits and lesioning methods.
Study authors, Nature
The study demonstrates that preventing the transient inflammatory response in grey matter blocks myelin regeneration in white matter. Conversely, when myelin fails to regenerate, the grey matter response becomes chronic, leading to sustained neuroinflammation and synapse loss — a pattern observed in neurodegenerative conditions.
Conversely, inducing myelin regeneration failure leads to chronic grey matter neuroinflammation. This recapitulates the low-grade inflammation considered to be a dominant mechanism underlying neurodegeneration.
Study authors, Nature
These findings indicate that white matter damage is not an isolated event but initiates a brain-wide repair response. When successful, this response restores myelin and resolves inflammation. When repair fails, the same protective mechanism turns harmful, driving persistent inflammation and cognitive decline.
Our findings reveal a form of regenerative plasticity coupling white matter integrity to grey matter function, which may underlie multiple neurodegenerative conditions and highlight the potential of targeting myelin regeneration to prevent chronic neuroinflammation.
Study authors, Nature
The research, conducted by scientists at the University of Cambridge and published in Nature, shifts focus from grey matter-centric models to the critical role of white matter in neurodegenerative disease progression. It suggests that therapies aimed at promoting myelin repair could interrupt the cycle that leads to chronic inflammation and synapse loss.
Previous understanding held that neurodegenerative diseases primarily involved direct damage to grey matter. This study shows instead that white matter injury can remotely affect grey matter through a regulated biological process — one that becomes pathological only when repair mechanisms fail.
The study’s authors emphasize that the observed microglial activation in grey matter should not be automatically interpreted as harmful. In the context of successful myelin regeneration, it serves a restorative purpose. Only when this process stalls does it contribute to disease.
By linking white matter integrity to grey matter health through a reparative pathway, the work offers a new framework for understanding how neurodegenerative diseases develop and progress. It also identifies myelin regeneration as a potential therapeutic target for conditions including Alzheimer’s disease, multiple sclerosis, and related disorders.
While the findings are based on animal models, they align with clinical observations of white matter lesions in patients with neurodegenerative diseases. The researchers note that further study is needed to determine how these mechanisms operate in humans and whether enhancing myelin repair can prevent or slow cognitive decline in clinical settings.
