
Yuhan Company’s Lecraza: A Landmark Approval for First Home Anti-Most cancers Drug Mixture Remedy within the US
The primary domestically produced anti-cancer drug accepted within the US… Third-generation lung most cancers therapy
Approval of Yuhan Recraza and Janssen Librivant mixture remedy
Preliminary therapy prescription for EGFR mutant non-small cell lung most cancers
30% ↓ danger of loss of life in comparison with normal Tagrisso therapy (monotherapy)
“Selling mixture remedy to maximise competitiveness via differential efficacy”
President Cho Wook-je: “We sit up for laying the muse for changing into a worldwide prime 50 pharmaceutical firm.”
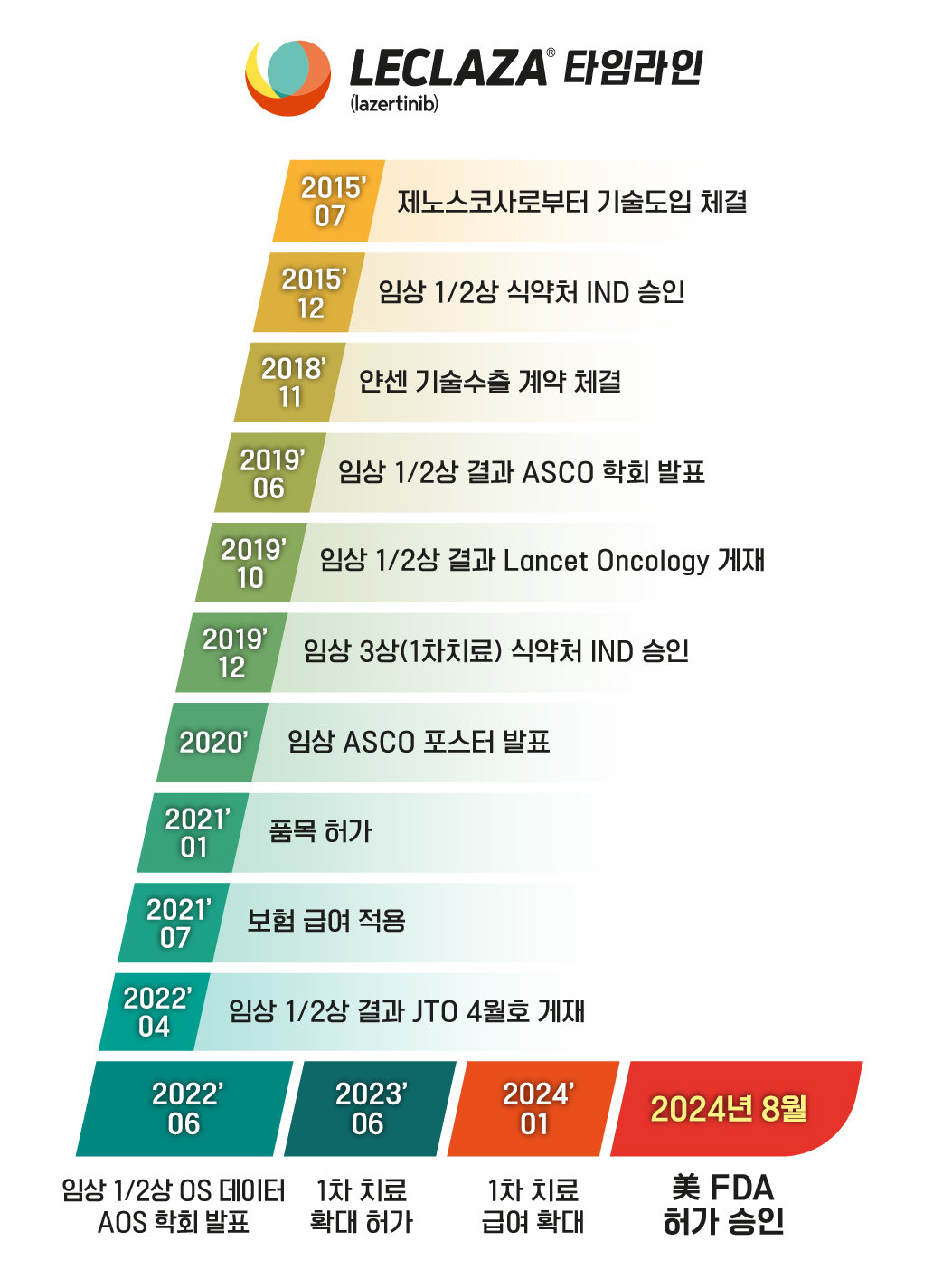
‘Lecraza’, the thirty first new Korean drug developed by Yuhan Company as a therapy for lung most cancers, obtained product approval as a mix remedy in the US. That is the primary time {that a} new home anticancer drug that isn’t biosimilar has been accepted in the US.
On the twentieth, Yuhan Company obtained approval from the US Meals and Drug Administration (FDA) for its third-generation lung most cancers drug ‘Lecraza (ingredient title: Lazertinib mesylate monohydrate, US product title: Rascluz)’ and the American focused bispecific antibody. the pharmaceutical firm Johnson & Johnson (J&J).
Mixture remedy of Lecraza and Librivant is initially really useful for grownup sufferers with domestically superior or non-small cell lung most cancers (NSCLC) with an Epidermal Development Issue (EGFR) exon 19 deletion. to substantiate or exon 21 substitution mutation (L858R). be used as a therapy.
Lecraza is a brand new third-generation anticancer drug developed to deal with EGFR mutation-positive non-small cell lung most cancers. A home pharmaceutical firm, a subsidiary of Oscotech (Genosco), developed Lazertinib and transferred the know-how to Yuhan Company within the preclinical stage, leading to Lecraza. In Korea, it’s used as a first-line monotherapy therapy for sufferers with domestically superior or non-small cell lung most cancers with EGFR mutations (exon 19 deletion and exon 21 substitution mutations). Paid prescriptions (1st and 2nd therapy) are additionally attainable.
Lung most cancers is a illness with a lot of sufferers worldwide and a excessive mortality charge, which makes it the most important anti-cancer drug market. As of final 12 months, the worldwide market dimension was mentioned to be round 46 trillion received. Relying on the scale of the most cancers cells, it’s divided into non-small cell lung most cancers and small cell lung most cancers (SCLC), and greater than 80% of all lung most cancers sufferers are identified with most cancers non-small cell lung. As well as, as genetic mutations equivalent to EGFR have been recognized in lung most cancers sufferers, remedies focusing on them have been developed. This may be understood as an idea of stopping the expansion of most cancers cells by eliminating all gene mutations that trigger lung most cancers. EGFR mutations are reported to happen comparatively often in Asians identified with non-small cell lung most cancers.
Generations of non-small cell lung most cancers therapy are divided by EGFR mutation and resistance efficacy. The third technology therapy was developed for the aim of treating sufferers who developed resistance after utilizing the primary and second technology remedies. Consultant examples embody the UK’s Tagrisso AstraZeneca (osimertinib, accepted by the US FDA in 2015) and Yuhan Company’s Lecraza (lasertinib).
Product picture of Recraza Yuhan Company This Recraza/Librivant mixture remedy was designated as a precedence evaluation goal by the FDA and has been reviewed. The approval was based mostly on the constructive outcomes of a section 3 scientific research (MARIPOSA) led by Janssen, a subsidiary of Johnson & Johnson. Janssen introduced that it confirmed that the Lecraza/Librivant mixture therapy diminished the danger of illness development or loss of life by 30% in comparison with AstraZeneca’s monotherapy Tagrisso (osimertinib). Key indicators embody development free survival (PFS) of 23.7 months, which is longer than Tagrisso (16.6 months), and length of response (DOR) of 25.8 months, which is roughly 9 months longer than osimertinib (16.8 months). Moreover, in a research on high-risk sufferers with confirmed TP53 mutations, mind metastases, and liver metastases, the mixed therapy group (27.5 months) confirmed constant PFS advantages in comparison with Tagrisso (18.5 months), and total survival (OS) additionally tends to be superior.
This approval is taken into account important as it’s the first non-chemotherapy case to exhibit superiority over current osimertinib, which is prescribed as a first-line therapy for EGFR-mutated NSCLC sufferers, and is the one multi-target mixture remedy.
Lecraza is a third-generation oral EGFR tyrosine kinase inhibitor (TKI) that’s extremely selective and might penetrate the mind. Right here, Librivant is a multi-target mixture remedy that maximizes the impact of the therapy by focusing on EGFR and the mesenchymal epithelial transition (MET) receptor, which has immune cell stimulating exercise.
Within the case of AstraZeneca, with a purpose to preserve Tagrisso’s competitiveness and market affect within the area of lung most cancers therapy, it carried out a scientific trial for a mix remedy combining Tagrisso and a chemotherapy drug. The outcomes additionally confirmed superior efficacy in comparison with Tagrisso monotherapy, and the drug was accepted as a first-line therapy by the FDA.
Evaluation means that the rationale for looking for product approval for Lazertinib as a mix remedy relatively than a monotherapy is as a result of it’s a latecomer. That is mentioned to be aimed toward differentiating its competitiveness via its larger therapeutic efficacy in comparison with Tagrisso, which was launched first and dominated the market. Actually, within the Lazertinib monotherapy scientific trial (LASER301), Lazertinib confirmed barely higher indicators in comparison with Tagrisso. Nevertheless, the PFS distinction is simply about 2 months, so it is onerous to say it is higher. General, efficacy and security are at related ranges. On this state of affairs, it’s analyzed {that a} mixture remedy has been strategically chosen to maximise the advantages in comparison with competing merchandise.
Yuhan Company evaluates that open innovation within the area of analysis and growth has had important outcomes with this FDA approval. Lecraza, which was accepted as a domestically produced new drug in January 2021, was lined by medical health insurance six months after its approval was prolonged to home first-line therapy in June final 12 months. Prescription gross sales totaled 20 billion received within the first quarter of this 12 months and are anticipated to exceed 100 billion received inside the 12 months. As well as, it’s anticipated that the approval of this mix remedy could have a constructive affect on approval critiques in different nations equivalent to Europe, China and Japan and the enlargement of home prescriptions.
Wook-Je Cho, CEO of Yuhan Company, mentioned, “The FDA approval of Recraza is a passing level, not an finish level,” and added, “We hope it is going to turn into a cornerstone for Yuhan Company to turn into a ‘prime worldwide. 50 pharmaceutical firms.”
Kim Min-beom, Donga.com reporter mbkim@donga.com
-
nice
0a canine -
I am unhappy
0a canine -
I am indignant
0a canine -
I like to recommend it
a canine
Sizzling information now
#Yuhan #Firms #lung #most cancers #drug #Recraza #domestically #produced #anticancer #drug #accepted #FDA #Mixture #remedy #accepted #firstline #therapy #DongA #Ilbo
