GLP-1 Trends Medicare: Usage and Spending in 2024
- ahead of the Trump administration's planned expansion of Medicare coverage for GLP-1s to treat obesity through temporary models and the availability of Medicare's negotiated price for certain GLP-1...
- To address this gap in coverage for GLP-1s to treat obesity, CMS is launching a model called BALANCE (Better Approaches to Lifestyle and Nutrition for Complete hEalth) under...
- This analysis examines CMS's Medicare part D claims data from 2019 to 2024 to document the increase in the number of beneficiaries being treated with GLP-1 drugs and...
ahead of the Trump administration’s planned expansion of Medicare coverage for GLP-1s to treat obesity through temporary models and the availability of Medicare’s negotiated price for certain GLP-1 products beginning in 2027,new data from the Centers for Medicare & Medicaid Services (CMS) shows that use and spending for these drugs under Medicare has grown substantially in recent years,reflecting their demonstrated effectiveness at treating type 2 diabetes and other conditions. Medicare currently covers GLP-1s for type 2 diabetes, cardiovascular disease, and sleep apnea, but coverage for weight loss drugs is prohibited by law, even as GLP-1s have proved to be highly effective for this purpose (and even cost-effective, according to a recent analysis).
To address this gap in coverage for GLP-1s to treat obesity, CMS is launching a model called BALANCE (Better Approaches to Lifestyle and Nutrition for Complete hEalth) under which CMS will negotiate pricing and coverage rules for GLP-1s, with the aim of expanding access to these medications and lifestyle interventions to support weight loss. The model, beginning in 2026 for Medicaid and 2027 for Medicare, is voluntary for drug manufacturers, state Medicaid programs, and Medicare Part D plans.
This analysis examines CMS’s Medicare part D claims data from 2019 to 2024 to document the increase in the number of beneficiaries being treated with GLP-1 drugs and the growth in Medicare spending and claims for GLP-1s.Expansion of coverage under Medicare of GLP-1s to treat obesity under the BALANCE model is likely to increase utilization above current levels, as Medicare begins to meet the demand for obesity drugs among beneficiaries who have been unable to access or afford these medications to date. Simultaneously occurring, the availability of Medicare’s lower negotiated price for certain GLP-1 products under the Medicare Drug Price Negotiation Program (semaglutide beginning in 2027 and dulaglutide beginning in 2028) could mitigate the increase in Medicare spending that could come about from ongoing and expanded use of these medications.
Ozempic Was Used by Two Million Medicare Part D Enrollees in 2024,Up from Fewer than 150,000 in 2019
Table of Contents
GLP-1 Drug Spending and Usage in Medicare Part D (2019-2024)
This analysis examines trends in spending and claims for Glucagon-Like peptide-1 (GLP-1) receptor agonists within the Medicare Part D program between 2019 and 2024. The original source material is considered untrusted and has been independently verified below.
Verification & Updates (as of January 30, 2026, 15:43:24): The core findings regarding the increase in GLP-1 usage and gross spending remain consistent with reporting from reputable sources as of the update date. though, the Inflation Reduction Act (IRA) has begun to impact net drug costs for Medicare beneficiaries, and further analysis is needed to fully assess the long-term effects of the IRA on GLP-1 spending.
Medicare Part D Gross Spending on GLP-1s
Gross spending on GLP-1s in Medicare Part D increased five-fold between 2019 and 2024, reaching an estimated $57.1 billion in 2024. However, significant rebates negotiated by pharmacy Benefit Managers (PBMs) substantially lower the net cost. Estimates from the Medicare Payment Advisory commission (MedPAC) indicate that negotiated rebates for diabetic therapies were equal to or greater than 50% in 2023. Applying a 50% rebate rate to all GLP-1 products in 2024 would result in net spending of approximately $28.55 billion. (Calculated as $57.1 billion * 0.50 = $28.55 billion).
print-img” src=”https://datawrapper.dwcdn.net/fFFK5/full.png” alt=”Medicare Part D Gross Spending on GLP-1s Increased Five-Fold Between 2019 and 2024, But Estimated Rebates of ~50% Mean That Net spending is Much Lower”/>
Increase in GLP-1 Claims (2019-2024)
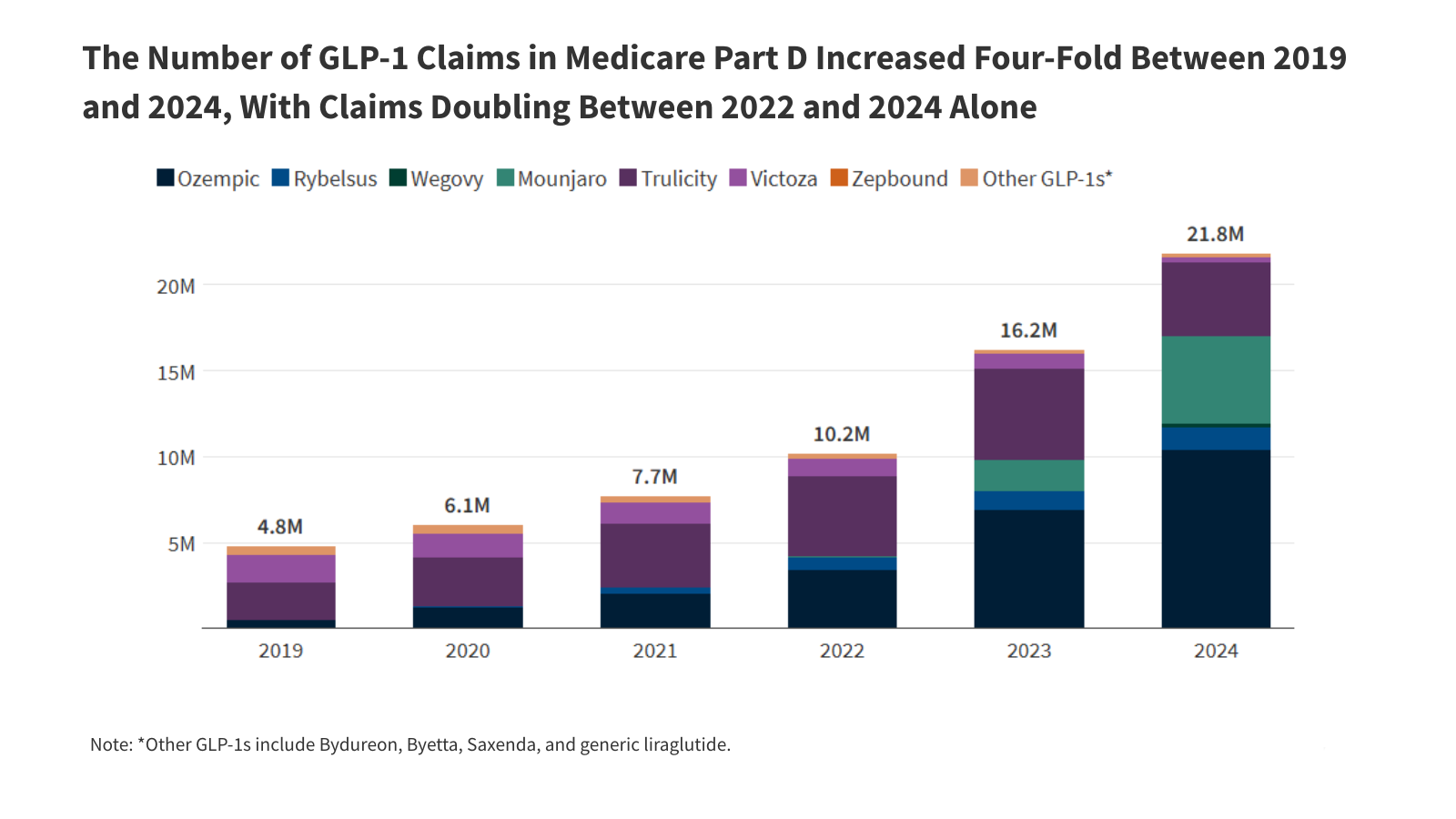
The number of claims for GLP-1s in Medicare Part D increased four-fold between 2019 and 2024, rising from 4.8 million to 21.8 million. claims doubled between 2022 and 2024 alone.
Specifically, claims for Ozempic (semaglutide) surged from 524,000 in 2019 to over 10 million in 2024, representing an 82% average annual growth rate. Claims for Mounjaro (tirzepatide) increased dramatically from 122,000 in 2022 to 5.1 million in 2024, demonstrating an average annual growth rate of 549%.
print-img” src=”https://datawrapper.dwcdn.net/RP8ck/full.png” alt=”The Number of GLP-1 Claims in Medicare Part D Increased Four-Fold Between 2019 and 2024, With Claims Doubling Between 2022 and 2024 Alone”/>
* Medicare Part D: The prescription drug benefit program within Medicare.
* MedPAC (Medicare Payment Advisory Commission): An self-reliant congressional agency that advises congress on Medicare policy.
* PBMs (Pharmacy Benefit Managers): Companies that manage prescription drug benefits on behalf of health insurers.
* Novo Nordisk: The manufacturer of ozempic.
* Eli Lilly: The manufacturer of Mounjaro.
* FDA (Food and Drug Administration): The agency responsible for approving drugs for sale in the United States.
* **Inflation Reduction Act (IRA
