Actin & Microtubules: How Cells Build Healthy Eggs | Northwestern Medicine
- The intricate process of egg cell development, a cornerstone of life itself, has been illuminated by new research from Northwestern Medicine.
- For an egg cell to fully develop, a group of specialized cells called nurse cells contribute their contents – all organelles and proteins – to the forming egg.
- Actin filaments and microtubules, while both components of the cytoskeleton, have traditionally been understood to play distinct roles.
The intricate process of egg cell development, a cornerstone of life itself, has been illuminated by new research from Northwestern Medicine. A study published in the Journal of Cell Biology details a previously unknown collaboration between actin filaments and microtubules – two essential components of a cell’s internal scaffolding – during the formation of developing egg cells. The findings, emerging from investigations conducted in fruit flies (Drosophila melanogaster), could have implications for understanding similar processes across species.
For an egg cell to fully develop, a group of specialized cells called nurse cells contribute their contents – all organelles and proteins – to the forming egg. This transfer of cellular material is a dramatic and fundamental process, yet the precise mechanisms governing it have remained elusive, according to Wen Lu, PhD, research assistant professor of Cell and Developmental Biology at Northwestern Medicine and a co-author of the study. “Exactly how What we have is accomplished has remained unclear,” she stated.
Actin filaments and microtubules, while both components of the cytoskeleton, have traditionally been understood to play distinct roles. Actin filaments are known for providing cells with flexibility and structural support, while microtubules function as more rigid “train tracks” for intracellular transport and maintaining cell shape. While the interaction between these two systems was already recognized, the specific coordination during complex developmental stages like egg formation was poorly defined. Vladimir Gelfand, PhD, the Leslie B. Arey Professor of Cell, Molecular, and Anatomical Sciences, and co-senior author of the study, explained that the mechanisms guiding this coordination remained largely unknown.
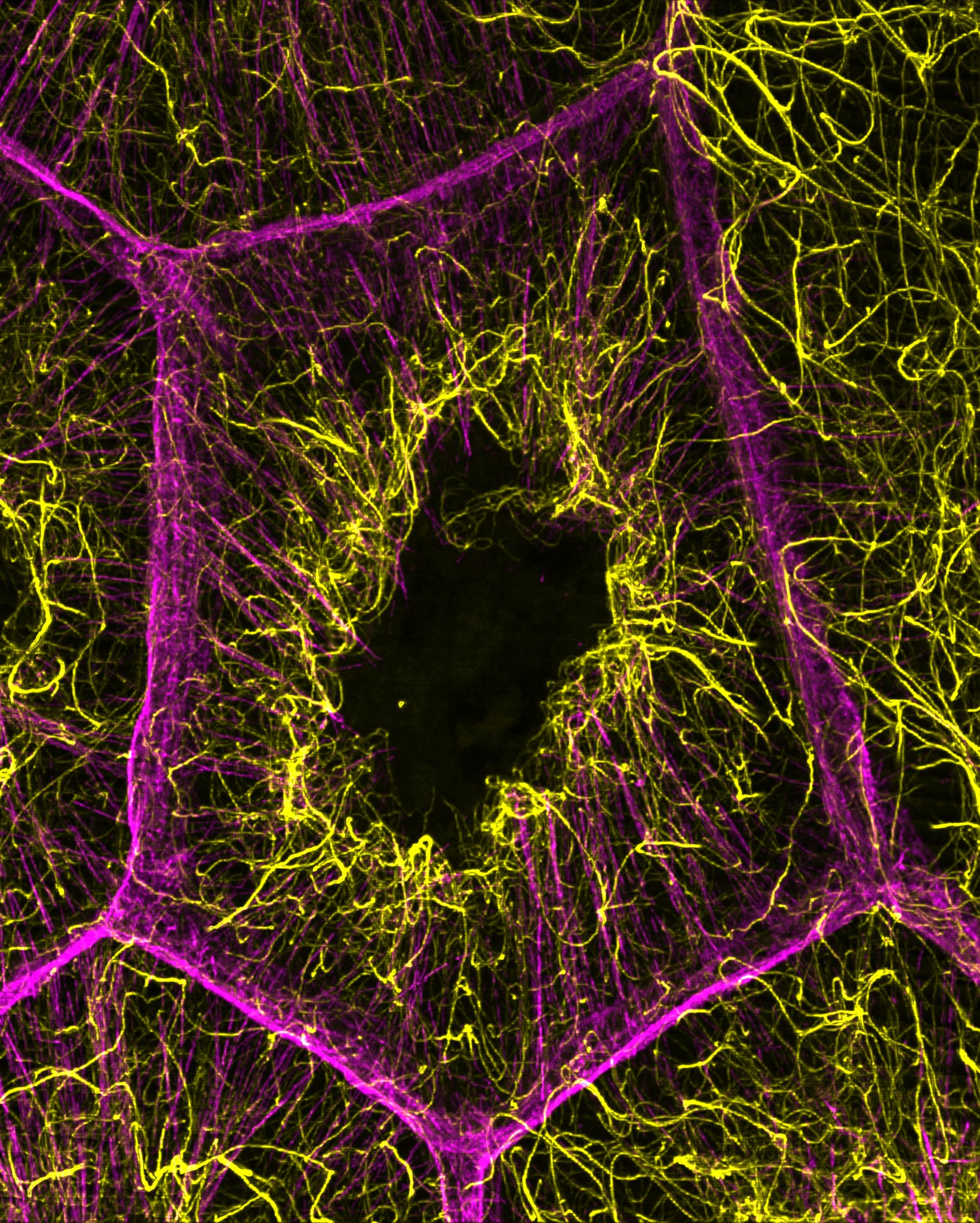
Using advanced microscopy techniques, the researchers observed that nurse cells build a stable network of acetylated microtubules concurrently with the formation of actin cables. Acetylation is a chemical modification that strengthens microtubules, and the study demonstrated its importance in maintaining this network. Disrupting the microtubule structure prevented both the initiation and elongation of actin cables, revealing that microtubules serve as a foundational scaffold during oogenesis – the process of egg cell development.
The research also revealed a reciprocal relationship: the actin cytoskeleton actively supports microtubule organization. Loss of key actin-bundling proteins led to fewer and shorter microtubules. This suggests that bundled actin filaments help regulate the spacing and orientation of microtubules, maintaining both stability and flexibility within the cell. This interplay highlights a complex “conversation” between the two systems, where each influences the assembly, positioning, and organization of the other, ultimately creating the architectural foundation necessary for transporting materials into the developing egg.
“This is a classic example of what we call ‘crosstalk’ in biology,” Gelfand said. “With this study, we now understand how these structures work together and are essential to oogenesis. Understanding this cytoskeletal coordination is essential, as many aspects of egg development are conserved across species.”
The study builds upon previous work by Gelfand’s team, published in September 2025, which revealed previously hidden dynamics within cells. That research, also utilizing advanced imaging, focused on the movement of vimentin intermediate filaments – key components of the cytoskeleton – within living cells. Traditionally considered static, these filaments were found to be highly mobile, traveling along microtubules. This earlier work underscored the idea that cellular structures previously thought to be rigid are, in fact, dynamic engines of cellular life.
Further research, published in December 2025, highlighted the role of actin in safeguarding chromosomes and microtubules during cell division. A study led by Yingqi Zhang at Zhejiang University demonstrated a new mitotic safeguard in early embryonic cells, reframing the understanding of this critical process. This research showed that actin, rather than solely relying on microtubules, plays a steering role in ensuring proper chromosome segregation.
Looking ahead, Gelfand, Lu, and their colleagues plan to utilize innovative microscopy techniques to visualize microtubules in even greater detail. “We are very lucky to have access to some of the best equipment in the world to help answer these fundamental scientific questions,” Lu said. The research was supported by National Institutes of Health grant R01-GM120378, National Institute of General Medical Sciences grant 2R35GM131752, and the CCBX Research Initiative from the Flatiron Institute/Simons Foundation.
The findings from Northwestern Medicine contribute to a growing body of knowledge regarding the intricate mechanisms governing cellular development. While the study was conducted in Drosophila, the fundamental principles of cytoskeletal coordination are likely conserved across species, offering potential insights into reproductive health and development in other organisms, including humans.
