Sleep Better, Breathe Easier: Alpibio’s Breakthrough Allergy Medicine Sees 28 Million Units Sold
- Alpibio announced on the 23rd that its domestically developed 'allergy medicine with minimal drowsiness side effect' had surpassed 28 million units in cumulative production in just 6 months...
- The recently launched general medicine 'Fexofenadine soft capsule allergy medicine' has officially surpassed 28 million units (in capsule units) in cumulative production in just 6 months since its...
- The production volume of this product has increased rapidly from 13 million units last year to 15 million units this year, achieving a 15.38% growth year-on-year.
Achieved 15.38% Growth: Korea’s First Allergy Medicine with Minimal Drowsiness Side Effect
allergy medicine with minimal drowsiness side effect’ (Fexofenadine soft capsule)” width=”100%” height=”auto” loading=”lazy” decoding=”async” />
Alpibio announced on the 23rd that its domestically developed ‘allergy medicine with minimal drowsiness side effect’ had surpassed 28 million units in cumulative production in just 6 months since its launch.
The recently launched general medicine ‘Fexofenadine soft capsule allergy medicine’ has officially surpassed 28 million units (in capsule units) in cumulative production in just 6 months since its domestic launch.
The production volume of this product has increased rapidly from 13 million units last year to 15 million units this year, achieving a 15.38% growth year-on-year.
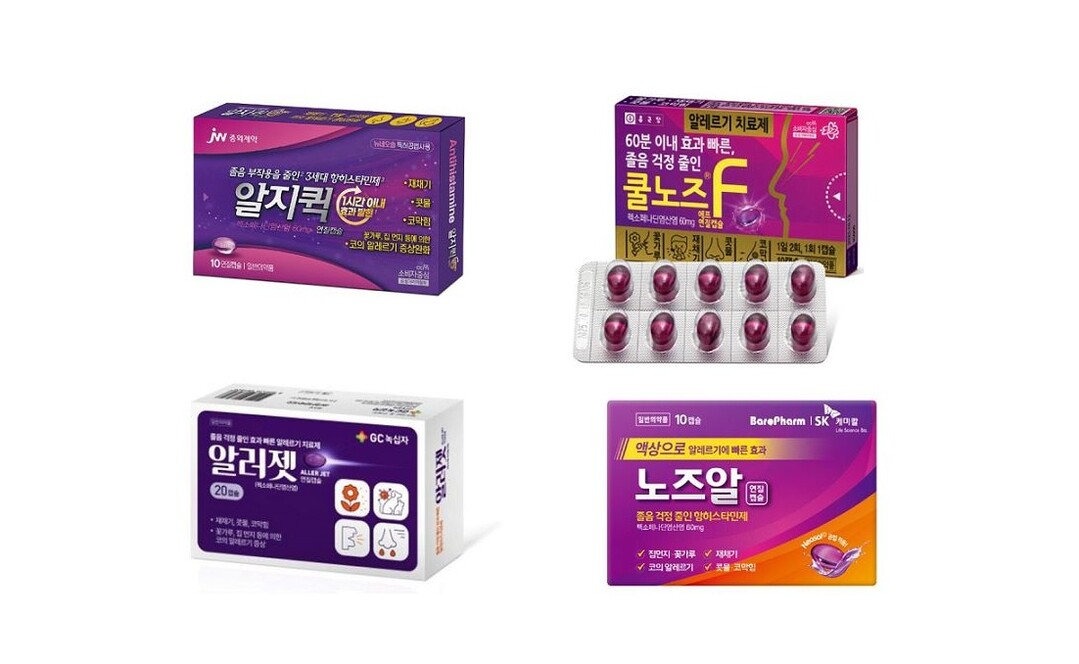
Currently, Alpibio is in charge of development and manufacturing, and four pharmaceutical companies are in charge of sales. The official distributors of this product are SK Chemicals (in partnership with Baropharm) ‘Nose Al Soft Capsule’, Green Cross ‘Allerjet Soft Capsule’, JW Pharmaceutical ‘Algiquick Soft Capsule’, and Chong Kun Dang ‘Cool Nose F Soft Capsule’.
Fexofenadine is the antihistamine with the least drowsiness side effect, and can be safely taken during the day. Alpibio has developed a soft capsule formulation of the fexofenadine ingredient from the existing tablet through Alpibio’s ‘Fexofenadine solubilization patented method’, which was developed for the first time in Korea, thereby improving the absorption rate and bioavailability.
This product is expected to be especially useful for consumers who need to avoid drowsiness during the day, such as students who need to focus on their studies and transportation workers for whom drowsy driving can lead to fatal injuries.
Fexofenadine is the only second-generation antihistamine that has been proven to be effective in alleviating allergic symptoms caused by pollen and house dust (indoor dust), and is therefore considered suitable as a first-line drug for allergic symptoms caused by fine dust in spring and fall.
