K-Medi Hub: Module for Molecular Site Diagnosis
- DAEGU, South Korea – K-Medi Hub (Daegu-Gyeongbuk Advanced Medical Industry Promotion Foundation) announced the development of a rapid diagnostic tool capable of quickly and accurately detecting VHSV, a...
- Researchers, including Park Ji-woong, head of the high-tech medical device development support center, and Lee Tae-jae, head of the Nano Technology Institute, collaborated with Fish Care (CEO Kim...
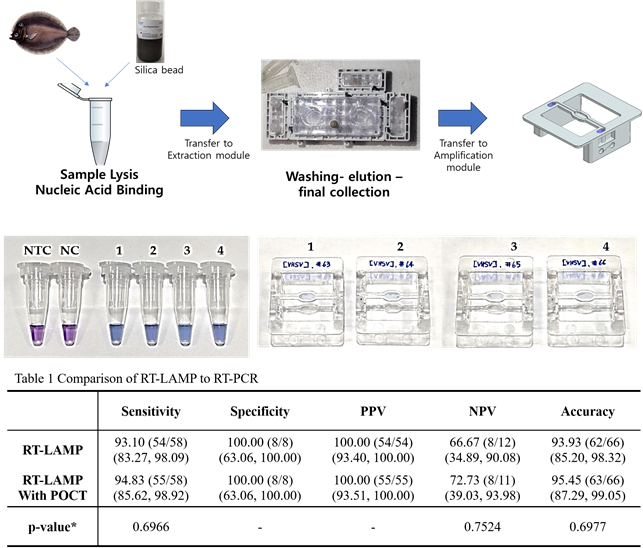
- The loop-mediated isothermal amplification (LAMP) technology employed in this research demonstrated superior performance compared to customary Point of Care Testing (PCR)-based diagnostics, according to K-Medi Hub.
K-Medi Hub Develops Rapid Virus Detection Technology
DAEGU, South Korea – K-Medi Hub (Daegu-Gyeongbuk Advanced Medical Industry Promotion Foundation) announced the development of a rapid diagnostic tool capable of quickly and accurately detecting VHSV, a virus, using the loop-mediated isothermal amplification (LAMP) method. The announcement was made by Park Gu-sun, Chairman of K-Medi Hub.
Field-Ready Diagnostic Platform
Researchers, including Park Ji-woong, head of the high-tech medical device development support center, and Lee Tae-jae, head of the Nano Technology Institute, collaborated with Fish Care (CEO Kim Seong-hyun) and Monitor (CEO Jeon Hyo-sung) to create the diagnostic equipment. The platform integrates viral nucleic acid extraction modules with miniaturized field molecular diagnostic modules, enabling immediate diagnostics in the field.
LAMP Technology Shows Promise
The loop-mediated isothermal amplification (LAMP) technology employed in this research demonstrated superior performance compared to customary Point of Care Testing (PCR)-based diagnostics, according to K-Medi Hub.
Publication in Biochip Journal
The findings of the research were recently published in the Biochip Journal, a Q1 journal ranking in the top 25% of journals in the ‘Chemistry’ and ‘Analytical’ categories within ‘Biochemical Research Methods’.
Supported by Government Funding
the project received support from the Ministry of Trade, industry and Energy and the Korea Institute of Industrial Technology Evaluation and Nano Technology.
Chairman’s Viewpoint
Park Gu-sun, chairman of K-Medi Hub, emphasized the meaning of the achievement. “This study is very meaningful in that it has secured core technology for in vitro diagnostic medical devices for field diagnosis,” Park said. “We expect that we can bring new vitality to the overall vitro diagnostic technology industry by advancing the technology of the infrastructure-based field diagnostic equipment.”
Rapid Virus Detection: A Q&A on K-Medi hub’s Groundbreaking Technology
K-Medi Hub has developed a rapid diagnostic tool for virus detection. Let’s delve into the details with some frequently asked questions.
What is K-Medi Hub?
K-Medi Hub (Daegu-Gyeongbuk Advanced Medical Industry Promotion Foundation) is the organization behind the development of this rapid virus detection technology. They are based in Daegu, South Korea.
what Virus Does This Technology Detect?
The technology is designed to quickly and accurately detect VHSV (Viral Hemorrhagic Septicemia Virus).
How Does the Rapid Virus Detection Technology Work?
The diagnostic tool uses the loop-mediated isothermal amplification (LAMP) method. LAMP technology allows for rapid amplification of viral genetic material, enabling swift detection. The platform integrates viral nucleic acid extraction modules with miniaturized field molecular diagnostic modules, allowing for immediate diagnostics in various environments.
What is LAMP Technology?
LAMP (loop-mediated isothermal amplification) is a method used to amplify a specific segment of DNA or RNA. It’s performed at a constant temperature, making it simpler and faster than customary PCR (Polymerase Chain Reaction) methods.A key advantage is its ability to be used in the field due to simpler equipment requirements.
What are the advantages of LAMP over PCR?
According to the information provided, LAMP technology demonstrated superior performance compared to PCR-based diagnostics, specifically Point of Care Testing. Advantages often include:
- Faster results
- Simpler equipment requirements
- Potentially more cost-effective in some situations
- Suitability for field use
Who Were the Key Collaborators in This Project?
The project was a collaborative effort involving several entities, including:
- Researchers from K-Medi Hub
- Nano Technology Institute (Led by Lee Tae-jae)
- Fish Care (CEO Kim Seong-hyun)
- Monitor (CEO Jeon Hyo-sung)
Where was the Research Published?
The findings of the research were published in the Biochip Journal.This is a Q1 journal, ranking in the top 25% of journals in the ‘Chemistry’ and ‘Analytical’ categories within ‘Biochemical Research Methods’.
Was This project Supported by Any funding?
Yes, the project received support from:
- The Ministry of Trade, Industry, and Energy
- The Korea institute of industrial Technology Evaluation and Nano Technology
What is the Meaning of This Technology, According to K-medi Hub?
Park Gu-sun, the chairman of K-Medi Hub, emphasized the significance of this achievement, stating that it secures core technology for in vitro diagnostic medical devices designed for field diagnosis. He expects this to bring “new vitality to the overall vitro diagnostic technology industry.”
What is “Field Diagnosis” and Why is it Crucial?
Field diagnosis refers to the ability to perform diagnostic tests outside of traditional laboratory settings. This is especially important for:
- Quickly identifying and containing outbreaks of infectious diseases.
- Providing rapid results in remote areas or situations where access to a lab is limited.
- Enabling on-the-spot testing for various conditions.
Why is Rapid Virus Detection Critically important?
Rapid virus detection is crucial for several reasons:
- Early Detection: Allows for timely intervention and treatment, potentially preventing the spread of the virus.
- Disease Control: Aids in the rapid identification of outbreaks, facilitating quarantine and other public health measures.
- Improved Patient Outcomes: Faster diagnosis can lead to quicker and more effective medical responses.
Comparing LAMP to PCR Detection Methods
Here’s a summarized comparison based on what we know from the article:
| Feature | LAMP (K-Medi Hub’s Technology) | PCR (Typical Point of Care Testing) |
|---|---|---|
| Performance | demonstrates superior performance. | Standard |
| Technology | Loop-mediated isothermal amplification | Polymerase Chain Reaction |
| Use Case | Field Diagnostics | Laboratory or specialized settings. |
This table provides a brief overview based on the provided article. Further research may be needed to provide more comprehensive comparisons.
This technology holds considerable promise for improving how we detect and respond to viral threats. The collaborative approach and the backing of government funding demonstrate the importance of this innovation.
